- Introduction
- Materials and Methods
- Chemicals and materials
- Isolation of tegument, membranes of host blood cells and lipids in host blood
- Incubation of adult worms and lipid extraction from culture supernatant and from hamster blood
- Lipid extraction
- Quantification and identification of phospholipid species composition
- Results
- PS species analysis
- PE species analysis
- PC species analysis
- PI species analysis
- Analysis of lysophospholipids
- Analysis of possible excretion of lysophospholipids by schistosomes
- Discussion
- Supplementary data
Chapter 6
The tegumental outer-surface membranes of Schistosoma mansoni are enriched in parasite-specific phospholipid species
*These authors contributed equally to this work
Affiliations
- F1
- Department of Biochemistry and Cell Biology, Faculty of Veterinary Medicine, Utrecht University, Utrecht, The Netherlands
- F2
- Department of Parasitology, Leiden University Medical Center, Leiden, The Netherlands
- F3
- Department of Medical Microbiology and Infectious Diseases, Erasmus MC, Rotterdam, The Netherlands
Abstract:
The complex outer-surface structure of adult Schistosoma mansoni, the tegument, is essential for survival of the parasite and consists of a syncytium of fused cells covered by two closely-apposed lipid bilayers that form the interactive surface with the host. In order to identify parasite-specific phospholipids present in the tegument, the species compositions of the major glycerophospholipid classes, phosphatidylcholine (PC), phosphatidylserine (PS), phosphatidylethanolamine (PE) and phosphatidylinositol (PI), including lysophospholipid species were analysed in adult S. mansoni worms, in isolated tegumental membranes and in hamster blood cells. It was shown that there are large differences in species composition in all four phospholipid classes between the membranes of S. mansoni and those of the host blood cells. The species compositions of PS and PC were strikingly different in the tegument compared to the whole worm. The tegumental membranes are especially enriched in lysophospholipids, predominantly eicosenoic acid (20:1)-containing lyso-PS and lyso-PE species. Furthermore, the tegument was strongly enriched in PC that contained 5-octadecenoic acid, an unusual fatty acid that is not present in the host. As we showed earlier that lysophospholipids from schistosomes affect the host-parasite interaction, excretion of these tegument-specific phospholipid species was examined in vitro and in vivo. Our experiments demonstrated that these lysophospholipids are not significantly secreted during in vitro incubations and are not detectable in peripheral blood of infected hosts. However, these analyses demonstrated a substantial decrease in PI content in blood plasma of schistosome infected hamsters, which might indicate that schistosomes influence exosome formation by the host.
Introduction
Schistosoma mansoni is a parasitic worm that causes schistosomiasis, a disease that afflicts over 200 million people and numerous animals in many rural areas in tropical countries (74). Adult S. mansoni worms are able to maintain themselves for many years in the blood vessels of their mammalian hosts (75). Despite their relatively large size (1 cm long with a diameter of 0.5 mm) compared to veins, and their abundant exposure to immune cells present in the blood of the host, the parasite apparently prevents clot formation and an adequate immune response of its host (108,115,137). Although the underlying molecular mechanisms involved in long-term parasite survival are not yet completely understood, the tegument outer-surface structure of schistosomes is of crucial importance for parasite survival and modulation of the host response (79,171).
The tegument of flatworms is a unique structure in nature that forms the interactive surface with the host. The tegument of schistosomes consists of a syncytium of fused cells covered by two closely-apposed lipid bilayers, constituting an inner plasma membrane and an outer membranocalyx (23,113,171). Multiple studies characterized the protein composition of these tegumental outer-surface membranes, which demonstrated that these membranes contain a specific set of proteins comprising nutrient transporters, structural membrane proteins, several proteins derived from the host and many proteins with a yet unknown function (15,31,184). The tegument of S. mansoni is also enriched in proteins that share no sequence similarity to any sequence present in databases of species other than schistosomes, demonstrating that the unique tegument structures comprise multiple unique proteins that are likely to fulfil schistosome-specific functions involved in parasite survival (15,171,184).
Next to proteins, the membranes of the tegument contain many distinct lipids and it was shown that eicosenoic acid (20:1), a rare species in the mammalian host, is a major component of the phospholipids of the tegumental membrane (2). The few early studies on the lipid compositions of the tegumental membranes provided little information on species compositions and were restricted by the limited technical possibilities of those days (2,151). More recent studies on tegumental membranes of schistosomes using MS techniques showed that the species composition of the phospholipids in the tegumental membranes differ not only substantially from those of blood cells of the host but also from those of whole adult worms (21,190). So far, the diacyl-phospholipid species composition of the tegument has been characterized of the two most abundant phospholipid classes in membranes of schistosomes, PC and PE (21). However, the species compositions of the less abundant phospholipid classes, such as phosphatidylserine (PS) and phosphatidylinositol (PI) have not yet been characterized in the tegument. These phospholipids are of interest as they are known to function as precursors for the production of several potent signalling molecules. Furthermore, schistosome lyso-PS (monoacylphosphatidylserine) species have been shown to activate Toll-Like Receptor 2 (TLR2) and affect dendritic cells of the host in such a manner that mature dendritic cells induce the development of IL-10 producing regulatory T cells, a process known to result in down regulation of the immune response (187). The present study provides a comprehensive analysis of the species compositions of all major glycerophospholipid classes, including PS, PI and lysophospholipids in isolated membranes of whole adult S. mansoni worms, in isolated tegumental membranes, and in membranes of host blood cells. In order to identify parasite-specific lipids the phospholipid species compositions in whole adult worms were compared to those of host blood cells. Furthermore, to identify phospholipid species enriched in the tegumental membranes, the phospholipid species compositions of tegumental membranes were compared to those of whole adult worms. Finally, the possible excretion into the environment of schistosome-specific phospholipids enriched in the tegumental membranes was investigated both in vitro and in infected hamsters.
Materials and Methods
Chemicals and materials
All solvents used were from Lab-Scan (Dublin, Ireland) and were of HPLC grade. Ammonium acetate, serine and Silica-60 for column chromatography were purchased from Merck (Darmstadt, Germany). Hydrochloric acid was purchased from Baker (St. Louis, MO, USA) and CM-52 was obtained from Serva (Heidelberg, Germany).
Isolation of tegument, membranes of host blood cells and lipids in host blood
Adult S. mansoni parasites were obtained by perfusion via the heart with 0.9% [w/v] NaCl of ether- or isofluorane-anesthetized hamsters at 45-49 days after infection. Isolated adult S. mansoni worms were washed in Hanks Balanced Salt Solution (HBSS) (161) before tegumental membranes were isolated by a freeze-thaw method according to Roberts et al. (1983)(147) as described by Brouwers et al. (1999)(19). Briefly, adult worms in HBSS were plunged drop by drop into liquid nitrogen. After thawing on ice, worms were extensively washed with ice-cold Tris-buffered saline (20 mM Tris-HCl, pH 7.4, 0.9% [w/v] NaCl, 1 mM of the protease inhibitor phenylmethylsulfonyl fluoride). The tegument membrane complex was removed from the worms by applying 10 vortex pulses of 1 sec. The supernatant, containing the tegumental membranes, was passed over a fine stainless-steel mesh and the filtrate was centrifuged at 5000g for 30 min at 4 °C. The isolated tegument membrane pellets were frozen at -20 °C until further use.
Blood was drawn from anesthetized hamsters by heart punctures. To determine the phospholipid composition of host blood cells, blood from non-infected hamsters was diluted (1:5, v/v) in water in order to lyse the red blood cells by hypotonic shock, shortly before lipid extraction. To analyse the phospholipids possibly excreted by schistosomes while living in the blood of the host, blood plasma from infected hamsters was prepared by centrifugation for 10 min at 1200g of citrated blood collected 45-49 days after infection with S. mansoni. As a control, plasma of non-infected hamsters was obtained by the same procedure.
Incubation of adult worms and lipid extraction from culture supernatant and from hamster blood
Adult S. mansoni worms were isolated by perfusion with warm (37 °C) 0.9% [w/v] NaCl and were then incubated for 2 h in S100 medium, which contained 20 mM Hepes (pH 7.4), 85 mM NaCl, 5.4 mM KCl, 0.7 Na2HPO4, 1 mM MgSO4, 1.5 mM CaCl2, 25 mM NaHCO3, 100mM glucose and 20% heat-inactivated human serum. After incubation the adult worms were removed from the medium and ice-cold tri-chloric acid (TCA) was added to the culture supernatant after which the proteins were allowed to precipitate on ice for 30 min. The culture supernatant was subsequently spun down at 18000g for 10 min. Lipids were extracted according to the method of Bligh and Dyer (1959) (12).
Lipid extraction
Lipids were extracted from the biological samples according to the method of Bligh and Dyer (1959) (12), with the minor modification that 6M HCl (0.1% v/v) was added at the second chloroform wash to increase recovery of acidic phospholipids. Neutral lipids were removed from the extracted lipids by fractionation on a 3 ml silica column prepared from 0.063-0.200 mm Silica 60. Lipid extracts were dissolved in chloroform and loaded on top of the silica column. Subsequently, neutral lipids were eluted with acetone (4 volumes) after which phospholipids and free fatty acids were eluted with methanol (4 volumes). This latter fraction was dried under nitrogen and stored at -20 °C until HPLC-MS analysis of the phospholipids.
Quantification and identification of phospholipid species composition
Isolated phospholipid samples were analyzed by an HPLC-MS method as previously described (145). In short, phospholipids were separated by HPLC, using a Synergi 4 μm MAX-RP 18A column (250 × 3 mm) (Phenomenex, CA, USA). Elution was performed within 55 minutes with a decreasing linear gradient of water in methanol/acetonitrile, with serine and ammonium acetate used as additives. Mass spectrometry was performed using electrospray ionization on a Sciex 4000QTRAP (Applied Biosystems, Nieuwerkerk aan de IJssel, the Netherlands). Samples were analyzed in a multiple reaction mode (MRM) or by neutral loss scanning or precursor scanning. Data analysis was performed according to the manufacturers' protocols with Markerview version 1.0 software (Sciex, Toronto, Canada).
Results
This study aimed to provide a comprehensive characterization of the glycerophospholipid and lysophospholipid species compositions of the tegumental membranes of adult S. mansoni worms, in order to identify parasite and tegument-specific phospholipids that might be involved in host-parasite interactions. We used a universal HPLC-mass spectrometry method for the identification of the molecular species of the four most abundant phospholipid classes (PC, PE, PS and PI) in homogenates of whole adult schistosomes, in isolated tegumental membranes of adult schistosomes and in blood cells of the host. The applied method separates isobaric species of all phospholipid classes prior to identification and quantification by mass spectrometry and therefore, allows detection and relative quantification of phospholipid species including lysophospholipids (145).
Tegumental membranes were isolated by a well-established freeze-thaw method originally developed by Roberts et al. (1983) (147). A modified version of this method has been used in our laboratory to characterize the proteome and the species compositions of the phospholipids PE and PC in the tegumental membranes of adult schistosomes (18,19,22,184).
Results of the current analysis of the species compositions of the analysed phospholipids in membranes of whole schistosomes (Table 6.1 and Tables S1-S3), are in general in agreement with our earlier study and the most abundant species of PS, PE, PC and PI in schistosomes were again found to be PS 18:0/22:4, PE 18:0/22:4, PC 16:0/20:1 and PI 18:0/22:4 (145). The present study did not focus on ether-linked PE species, because the positive ion mode now used is less suitable for detection of these PE phospholipids (23). Therefore, this study could not confirm the previously observed abundant presence of ether-linked PE species in schistosome membranes nor the specific enrichment of ether-linked PE species in tegumental membranes, such as 16:0/20:1 plasmalogen-PE (21).
PS species analysis
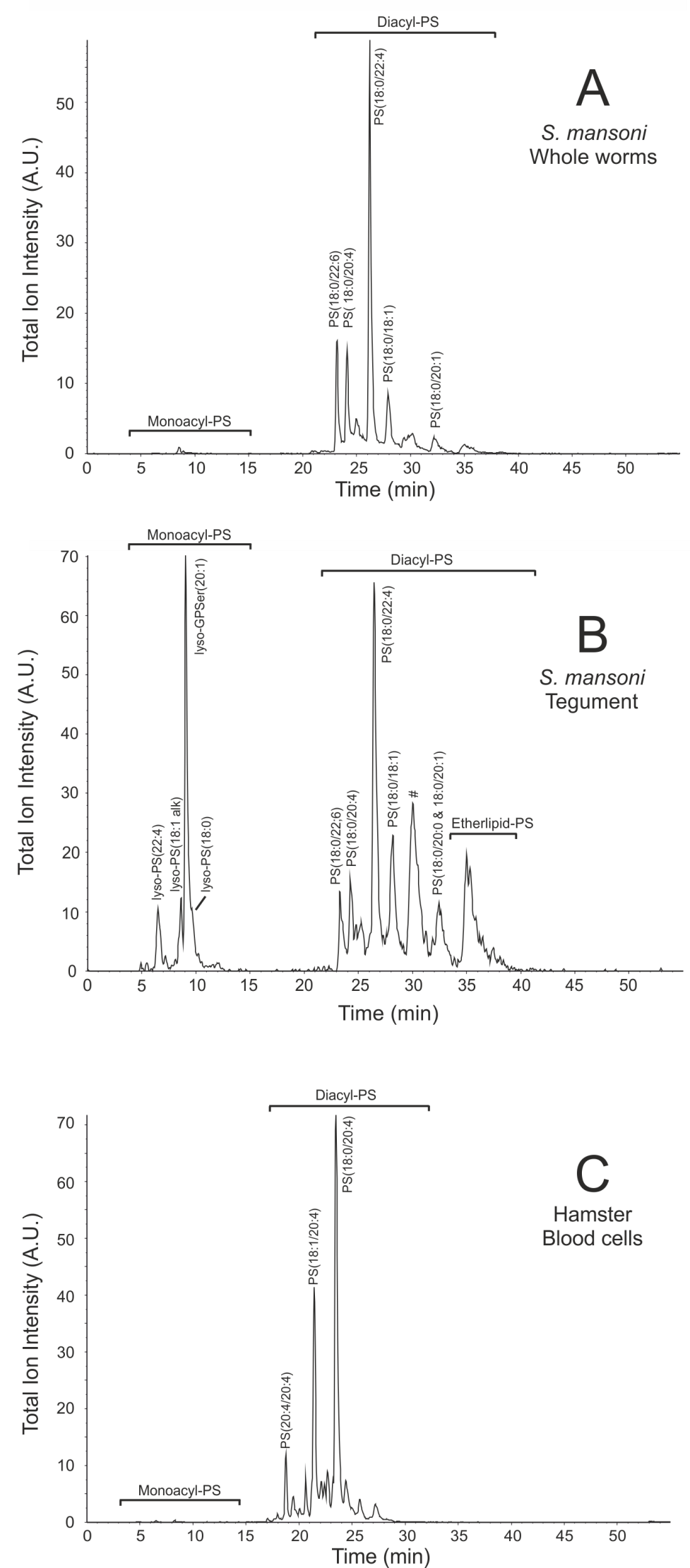
The HPLC-elution pattern of PS species derived from tegumental membranes differed drastically from that of membranes of whole worms and from that of membranes from blood cells, as the tegumental membranes clearly contain much more lyso-PS and etherlipid-PS (Fig. 6.1). Identification and quantification of the PS species demonstrated that schistosome membranes comprise many distinct PS species hardly present in the membranes of blood cells, such as PS 18:0/22:4, PS 18:0/20:1 and several etherlipids (Table 6.1). PS species containing a fatty acid of 22 or more carbon atoms were abundantly present in schistosomal membranes when compared to membranes of blood cells, 57% versus 9% respectively (Table 6.1). On the other hand, species with 22 or more carbon atoms are much less present in the tegument when compared to membranes of whole worms and comprise only 28% of the PS species in the tegument (Table 6.1).
In order to identify the PS species most different in abundance between membranes of adult worms, the tegument and blood cells, a discriminant analysis was performed. This statistical method identifies the components that differ most between groups of samples and thus contribute most to the distinction between these samples. This discriminant analysis identified lyso-PS 20:1 (m/z = 550), lyso-PS 22:4 (m/z = 572) and the PS species with an m/z ratio of 772, 800, 804, 830 and 832 as the PS species most abundant in the tegumental membranes (Fig. S1). At least 4 of th ese 5 PS species contain ether-linked acyl chains (Table 6.1). Therefore, the PS species composition in the tegumental membranes is specifically enriched in ether-linked PS species (Fig. 6.1), which is similar to the already reported enrichment of ether-linked PE species in the tegument (21).
| Whole worm (n=5) |
Tegument (n=3) |
Hamster blood cells (n=5) |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| m/z | Rt | Acyl chains | Component | % | ± | S.D. | % | ± | S.D. | % | ± | S.D. | |
| 760 | 24.7 | 16:0/18:1 | diacyl PS | * | * | 1.0 | ± | 0.1 | |||||
| 772 | 29.7 | 16:0/20:1 | plasmalogen PS | 1.9 | ± | 0.2 | 5.2 | ± | 1.4 | * | |||
| 782 | 21.7 | 16:0/20:4 | diacyl PS | * | * | 1.8 | ± | 0.2 | |||||
| 784 | 22.7 | 18:1/18:2 | diacyl PS | * | * | 1.3 | ± | 0.1 | |||||
| 786 | 25.1 | 18:0/18:2 | diacyl PS | 2.6 | ± | 0.2 | 1.5 | ± | 0.1 | 2.5 | ± | 0.1 | |
| 788 | 28.5 | 18:0/18:1 | diacyl PS | 5.3 | ± | 0.5 | 3.2 | ± | 0.5 | 2.4 | ± | 0.3 | |
| 796 | 22.8 | 17:0/20:4 | diacyl PS | * | * | 1.9 | ± | 0.1 | |||||
| 798 | 31.4 | # | 1.6 | ± | 0.2 | 3.8 | ± | 1.2 | * | ||||
| 800 | 35.8 | 18:0/20:1 | plasmalogen PS | 3.6 | ± | 0.6 | 10.1 | ± | 2.9 | * | |||
| 802 | 27.6 | # | * | 2.0 | ± | 0.7 | * | ||||||
| 804 | 29.6 | 38:0 | alkyl,acyl PS | * | 7.1 | ± | 0.7 | * | |||||
| 806 | 19.9 | 18:2/20:4 | diacyl PS | * | * | 2.4 | ± | 0.2 | |||||
| 808 | 22.0 | 18:1/20:4 | diacyl PS | * | * | 14.8 | ± | 0.6 | |||||
| 810 | 24.1 | 18:0/20:4 | diacyl PS | 5.4 | ± | 0.3 | 2.3 | ± | 0.3 | 36.0 | ± | 2.7 | |
| 812 | 25.6 | 18:0/20:3 | diacyl PS | 2.0 | ± | 0.1 | * | 4.8 | ± | 0.3 | |||
| 814 | 28.1 | 18:0/20:2 | diacyl PS | 1.9 | ± | 0.1 | 1.3 | ± | 0.2 | * | |||
| 816 | 32.1 | 18:0/20:1 | diacyl PS | 3.5 | ± | 0.4 | 5.4 | ± | 0.3 | * | |||
| 818 | 32.1 | 18:0/20:0 | diacyl PS | * | 1.8 | ± | 0.1 | * | |||||
| 822 | 29.0 | # | 1.3 | ± | 0.2 | 1.4 | ± | 0.2 | * | ||||
| 824 | 25.3 | 19:0/20:4 | diacyl PS | * | * | 1.1 | ± | 0.1 | |||||
| 830 | 19.2 | 20:4/20:4 | diacyl PS | * | * | 4.4 | ± | 0.5 | |||||
| 830 | 29.8 | # | * | 4.1 | ± | 0.5 | * | ||||||
| 832 | 22.0 | 40:7 | diacyl PS | * | * | 3.0 | ± | 0.1 | |||||
| 832 | 35.0 | 40:0 | alkyl,acyl PS | * | 9.7 | ± | 1.4 | * | |||||
| 834 | 22.0 | 18:1/20:5 | diacyl PS | * | * | 1.9 | ± | 0.1 | |||||
| 834 | 23.2 | 18:0/22:6 | diacyl PS | 7.6 | ± | 1.3 | 4.2 | ± | 0.2 | 3.5 | ± | 0.3 | |
| 836 | 24.1 | 18:1/22:4 | diacyl PS | * | * | 1.9 | ± | 0.1 | |||||
| 836 | 25.2 | 18:0/22:5 | diacyl PS | 5.2 | ± | 0.5 | 3.0 | ± | 0.9 | 1.2 | ± | 0.1 | |
| 838 | 26.1 | 18:0/22:4 | diacyl PS | 34.3 | ± | 1.8 | 16.5 | ± | 2.2 | 2.8 | ± | 0.2 | |
| 840 | 28.9 | # | 5.3 | ± | 0.2 | 2.6 | ± | 0.4 | * | ||||
| 854 | 27.9 | # | * | 1.6 | ± | 0.5 | * | ||||||
| 864 | 27.0 | # | 1.7 | ± | 0.2 | * | * | ||||||
| 866 | 29.4 | 20:0/22:4 | diacyl PS | 1.1 | ± | 0.1 | * | * | |||||
| 866 | 30.0 | 18:0/24:4 | diacyl PS | 1.4 | ± | 0.1 | * | * | |||||
PE species analysis
Analysis of the species compositions of PE showed striking differences between PE in blood cells and in lipids of schistosomes (Table S1). The three most abundant species in PE of hamster blood cells (18:1/20:4, 18:1/18:2 and 16:0/18:1) are not even among the top 10 of the most abundant PE species in worms. The reverse is also true: the most abundant PE species in schistosomes (18:0/22:4, 18%) ranks as number 15 in PE species of hamster blood cells and is only present in trace amounts (1.2%).
In contrast to the large difference in PS species composition between whole worms and the tegument, there were no significant differences observed in PE species of the tegument compared to the whole worm (Table S1).
PC species analysis
The species composition of PC in schistosomes was clearly different from that of hamster blood cells (Table S2). For example, the number one in PC of the worms with 14% was 16:0/20:1, while that species represented less than 1% of the total PC in hamster blood cells. The most abundant PC species in hamster blood cells was 16:0/18:2 (20%), while this species ranked 5th and represented only 5% of the total PC species in the worms.
Striking, however, are also the differences in PC species composition between worms and their tegumental membranes. The tegument is twofold enriched in 16:0/16:0 diacyl PC, the most abundant PC species in these membranes (Table S2). The most remarkable difference between tegument and worms is the gross enrichment in 16:0/18:1 PC, where the 18:1 fatty acid is very unusual as we showed earlier that it contains the double bond at C5, instead of the common C9 position (22). In tegumental membranes this 16:0/18:1 5 PC represented 12% of the total PC, while worm PC contained only 2%. This is in agreement with our earlier study where we showed that this rare PC species is not detectable in blood of the host and is, apart from dipalmitoyl PC, the most abundant PC species in the tegument of schistosomes (22).
PI species analysis
Analysis of the species composition of PI revealed that also large differences existed between the PI species composition of blood cells and that of the schistosome membranes (Table S3). Blood cells contain predominantly PI 18:0/20:4 and PI 18:0/18:2, representing together half of the PI species in hamster blood cells. On the other hand, membranes of whole worms as well as tegumental membranes contained in addition to PI 18:0/20:4, a large amount of PI 18:0/18:1 (21 and 27% in worms and tegument respectively), which represents in hamster blood cells only 2% of the total PI species (Table S3).
When compared to the differences in species compositions of PC and PS, the differences we observed in PI species composition are remarkably small between whole worms and tegumental membranes. PI species are well known to generate potent signaling molecules, but the absence of specific enrichment of certain PI species in the tegument, suggests that specific PI species are not involved in tegument-specific functions.
Analysis of lysophospholipids
Next to analysis of the diacyl-phospholipid species, we also investigated the presence of monoacyl-phospholipid (lyso-PL) species of the four above mentioned phospholipid classes, as it had been reported that lyso-PC as well as lyso-PS species are important in host-parasite interactions in schistosomiasis (69,106,187). Lysophospholipids and diacyl-phospholipids do not ionize with the same efficiency, and therefore, these phospholipids are not detected with the same efficiency by mass spectrometry. For this reason, the molar ratio between lysophospholipids and diacyl-phospholipids cannot be estimated by mass spectrometry unless careful calibration is performed with a large array of standards, which are not commercially available. However, in order to determine whether one of the three investigated membrane fractions contains a relatively large proportion of lysophospholipid species, the ratio of the detected signals of lysophospholipid species over that of diacyl-phospholipid species can be compared between these fractions. Table 6.2 shows the ratio of detected signals derived from lysophospholipid species over that of diacyl-phospholipids for each phospholipid class in the three analyzed membrane preparations. These results are in line with earlier observations that the schistosomula of S. mansoni can produce lyso-PC (71), and we now show that in adult schistosomes the tegumental membranes, when compared to whole worms, are enriched in lyso-phospholipid species, especially lyso-PS and lyso-PE.
| Whole worms (n=5) |
Tegument (n=4) |
Hamster blood cells (n=4) |
|||||||
|---|---|---|---|---|---|---|---|---|---|
| ratio | ± | S.D. | ratio | ± | S.D. | ratio | ± | S.D. | |
| lyso-PS/diacyl-PS | 1.0 | ± | 0.2 | 15.8 | ± | 4.0 | 2.3 | ± | 2.9 |
| lyso-PE/diacyl-PE | 1.0 | ± | 0.3 | 7.2 | ± | 7.1 | 1.3 | ± | 0.4 |
| lyso-PC/diacyl-PC | 1.0 | ± | 0.4 | 1.6 | ± | 0.9 | 1.4 | ± | 0.5 |
| lyso-PI/diacyl-PI | 1.0 | ± | 0.1 | 3.4 | ± | 0.2 | 0.2 | ± | 0.1 |
Analysis of the lysophospholipid species of the distinct phospholipid classes demonstrated that in blood cells the total acyl-chain length and number of desaturations of the minor amounts of lyso-PC, lyso-PE and lyso-PS were very similar to each other, because over 75% of the lysophospholipid species in these classes comprised acyl chains with 16 or 18 carbon atoms and none or a single desaturation (not shown). The lyso-PI species composition in blood cells also contained a large amount of lyso-PI 20:4 in addition to the lyso-PI species with acyl chains of 16 and 18 carbon atoms (not shown). On the other hand, membranes of adult S. mansoni worms had a lyso-phospholipid species composition with species containing unsaturated acyl chains of 16 or 18 carbon atoms and substantial amounts (over 20%) of lyso-PS 20:1 and lyso-PE 20:1 (Table 6.3). Interestingly, these two lysophospholipid species were drastically enriched in the tegumental membranes when compared to whole worms, and these species were by far the most abundant lyso-PS and lyso-PE species present in the tegumental membranes (Table 6.3). Eicosenoic acid (20:1) is synthesized by the parasite by chain elongation of oleic acid (18:1) and it is an abundantly present fatty acid in schistosomes while virtually absent in the host (18,20,68). The enrichment in the tegument of eicosenoic acid containing lyso-PS and lyso-PE is not observed in lyso-PC and lyso-PI, and the species composition of these lysophospholipid classes is dominated by species containing unsaturated acyl chains of 16 or 18 carbon atoms in lyso-PC and lyso-PI, respectively (Table 6.3). Therefore, no major differences exist in the lysophopholipid species composition of lyso-PC and lyso-PI between tegumental membranes and those whole worms, whereas the tegumental membranes are drastically enriched in lyso-PE and lyso-PS species containing eicosenoic acid (20:1).
| Fatty acyl | Lyso PS (% ± S.D.) | Lyso PE (% ± S.D.) | Lyso PC (% ± S.D.) | Lyso PI (% ± S.D.) | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| m/z | Worms (n=5) | Teguments (n=5) | m/z | Worms (n=5) | Teguments (n=5) | m/z | Worms (n=5) | Teguments (n=5) | m/z | Worms (n=5) | Teguments (n=5) |
|||||||||||||||||
| 16:0 | 496 | 1.5 | ± | 0.3 | 2.5 | ± | 1.1 | 454 | 7.1 | ± | 4.7 | 7.7 | ± | 3.3 | 496 | 38.4 | ± | 4.0 | 47.8 | ± | 3.1 | 571 | 1.8 | ± | 0.9 | 5.6 | ± | 1.1 |
| 18:1 alk | 508 | 7.6 | ± | 2.3 | 4.1 | ± | 3.8 | 466 | * | * | 508 | * | * | 583 | * | * | ||||||||||||
| 18:0 alk | 510 | 2.1 | ± | 0.6 | * | 468 | * | * | 510 | * | * | 585 | 5.3 | ± | 0.6 | 2.6 | ± | 1.1 | ||||||||||
| 18:2 | 520 | * | * | 478 | * | * | 520 | 2.4 | ± | 1.0 | 2.6 | ± | 1.8 | 595 | * | 4.5 | ± | 0.8 | ||||||||||
| 18:1 | 522 | 1.3 | ± | 0.7 | 1.5 | ± | 0.4 | 480 | 8.3 | ± | 4.2 | 11.5 | ± | 2.2 | 522 | 9.1 | ± | 1.7 | 11.4 | ± | 3.1 | 597 | 3.2 | ± | 2.0 | 11.7 | ± | 3.6 |
| 18:0 | 524 | 46.2 | ± | 4.4 | 9.3 | ± | 4.2 | 482 | 63.1 | ± | 8.5 | 19.8 | ± | 9.5 | 524 | 27.5 | ± | 2.2 | 23.9 | ± | 5.9 | 599 | 80.2 | ± | 4.3 | 62.8 | ± | 4.8 |
| 20:4 | 544 | * | * | 502 | * | * | 544 | 2.6 | ± | 0.3 | * | 619 | 1.3 | ± | 0.4 | 3.1 | ± | 1.0 | ||||||||||
| 20:2 | 548 | * | * | 506 | * | * | 548 | 3.6 | ± | 1.2 | 3.1 | ± | 1.3 | 623 | * | 1.5 | ± | 0.9 | ||||||||||
| 20:1 | 550 | 24.0 | ± | 4.5 | 62.6 | ± | 7.2 | 508 | 21.6 | ± | 10.4 | 57.1 | ± | 8.1 | 550 | 9.0 | ± | 1.1 | 8.0 | ± | 3.6 | 625 | * | 2.0 | ± | 0.9 | ||
| 20:0 | 552 | 2.1 | ± | 0.4 | 2.6 | ± | 0.4 | 510 | * | 1.4 | ± | 2.2 | 552 | 2.3 | ± | 0.3 | * | 627 | * | * | ||||||||
| 22:4 | 572 | 8.1 | ± | 1.4 | 10.1 | ± | 0.7 | 530 | * | * | 572 | 1.3 | ± | 0.7 | * | 647 | * | * | ||||||||||
| 22:1 | 578 | * | 1.3 | ± | 0.7 | 536 | * | * | 578 | * | * | 653 | * | * | ||||||||||||||
| 24:1 | 606 | 1.3 | ± | 0.2 | * | 564 | * | * | 606 | * | * | 681 | 5.2 | ± | 1.4 | 4.4 | ± | 1.2 | ||||||||||
Analysis of possible excretion of lysophospholipids by schistosomes
Because of the increased watersolubility of lysophospholidpids compared to diacyl phospholipids, the specific enrichment of lysophospholipids in the tegumental membranes that form the outer-surface of the parasite, and because of the suggested function of lysophospholipids in host-parasite interactions (106,146,187), we investigated whether these lysophospholipids are excreted into their environment. Lysophospholipid secretion by schistosomes was examined in vitro by total phospholipid analysis of medium incubated with or without adult schistosomes. None of the schistosome-specific lysophospholipid or phospholipid species appeared to be present in the medium incubated with schistosomes, and no significant differences in species composition of PC, PE, PS and PI were detected between medium incubated with schistosomes and control medium (not shown).
The possible excretion of tegument specific lysophospholipids was also investigated in vivo by analysis of total phospholipids present in blood plasma of infected and non-infected hamsters. Again none of the schistosome-specific or tegument-specific lysophospholipids or diacylphospholipid species was increased in plasma derived from schistosome infected hamsters when compared to non-infected hamsters. These results demonstrate that the tegument-specific lyso-phospholipids are not significantly secreted into aqueous solutions with albumin and do not circulate in blood of infected hosts. However, a substantial decrease in total PI content was detected in plasma from infected hamsters compared to non-infected hamsters (Table 6.4). All these 5 PI species are common and abundant diacyl PI species in host blood cells (Table S3).
| m/z | PL class | Acyl chains | Ratio infected/control | P-value |
|---|---|---|---|---|
| 833 | PI | 34:2 | 0.35 | 1.17 10-7 |
| 861 | PI | 36:2 | 0.37 | 5.46 10-6 |
| 857 | PI | 36:4 | 0.38 | 1.21 10-6 |
| 887 | PI | 38:3 | 0.40 | 4.96 10-5 |
| 885 | PI | 38:4 | 0.42 | 2.61 10-6 |
Discussion
The tegument outer-surface membranes of adult schistosomes, a unique biological structure which forms the site of interaction with the host (113), comprises many schistosome-specific and tegument-specific lipids. In addition to the already reported tegument-specific enrichment of several PC and ether-linked PE species compared to whole worms (18,22), our results showed that also multiple PS species are specifically enriched in the tegument. On the other hand, no tegument-specific PI species could be detected. The species composition of the most abundant phospholipid classes (PC and PS) in tegumental membranes differs drastically from that of whole worms. However, this phenomenon is not true for all phospholipid classes, as the species compositions of PI and PE in tegumental membranes did not differ from those of whole worms. Furthermore, the enrichment in the tegument of eicosenoic acid (20:1) containing lysophospholipids was only observed for PS and PE and not for PC and PI. These results, therefore, suggest that the tegument comprises mechanisms that facilitate the enrichment of certain phospholipids in the membranes of the tegument. The function of most of the identified tegument-specific phospholipids and lysophospholipids is not yet known, but the lyso-PS and lyso-PC species of schistosomes have been shown to activate TLR-2 on host immune cells, resulting in a down-regulation of the host immune response and eosinophil activation and recruitment, respectively (106,146,187).
Interestingly, the various phospholipid species enriched in the tegument of the distinct phospholipids classes do not contain a similar set of acyl chains, as different species for PC and PS are enriched in the tegument (Table 6.1 and supplementary data). Hence, the tegumental membranes are enriched in specific phospholipid species that differ between the distinct phospholipid classes.
Earlier observations on a different life cycle stage, schistosomula, clearly demonstrated that this stage exports home made lyso-phospholipids (69). It is unknown yet whether adult schistosomes excrete the schistosome-specific and/or tegument-specific phospholipids. Detection of the possible excretion of these lipids is hampered by the biophysical properties of the lipids in question, as they contain relatively hydrophobic long-chain fatty acids and lysophospholipids that can be expected to partition quickly into nearby phospholipid membranes. In vivo, the tegument-specific lysophospholipids might spread quickly to nearby membrane structures, which would then allow receptor activation of host immune cells as reported in in vitro studies (106,146,187). Knowing this, we nevertheless searched specifically for excretions of those schistosome-specific lipids by adult schistosomes. We could, however, not detect any significant secretion of these lipids by schistosomes, not during our in vitro incubations, nor in the blood of infected hamsters. Therefore, the in vivo mechanism by which these lysophospholipids might fulfil the in vitro observed effects on host immune cells remains to be resolved and probably requires sophisticated co-culture studies that are beyond the scope of this study.
However, analysis of the phospholipid composition in blood plasma of infected and non-infected hamsters demonstrated another interesting difference, as a substantial decrease in PI content was observed in blood plasma of schistosome infected hamsters. As phospholipids are not bound to albumin, because this protein binds predominantly fatty acids, these phospholipids probably constitute small vesicular structures, such as exosomes. The substantial decrease in PI is peculiar as this phospholipid is well known for its precursor function in signal transduction. Therefore, these results might indicate that the schistosome infection alters exosome formation by endothelial cells or by circulating immune cells, which is under current investigation.
Supplementary data
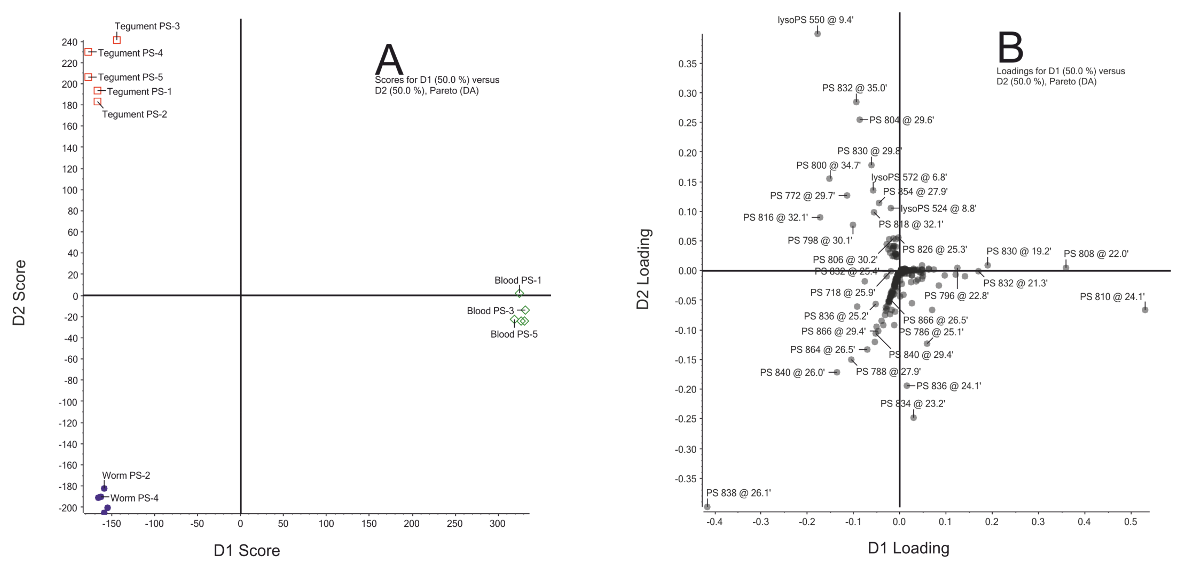
This discriminant analysis revealed that the differences among the independent replicates of the three investigated membrane preparations (whole worms, tegument and host blood cells), were much smaller than the differences between the three distinct membrane preparations (Fig. S1A). Discriminant 1 (represented by the X-axis in panel A) indicates the differences in PS species between blood cells and those of schistosomes, whereas discriminant 2 (represented by the Y-axis in panel A) indicates the differences in PS between tegumental membranes and those of whole adult worms. In the corresponding loading plot (panel B) all PS species are represented by a dot and those PS species that do not vary in abundance between the distinct membrane preparations are plotted close to the origin of the X-axis and Y-axis, as they hardly contribute to the distinction between the three membrane preparations. On the other hand, the PS species that vary in abundance between blood cells and those of schistosomes are plotted far from the origin of the X-axis. For instance, PS 18:0/20:4 and PS 18:1/20:4 with a m/z ratio of 810 and 808, respectively, are the two most abundant PS species in blood cells and present in very low amounts in both schistosome samples (Table 6.1). Hence, these species are major discriminating PS species between blood cells and schistosome samples, and therefore, these PS species are located in the loading plot of the discriminant analysis at a position with a high loading on discriminant 1 (Fig. S1B, on the X-axis, far right in bottom corner). Similarly, PS 18:0/22:4 (m/z = 838), which is abundantly present in lipid samples of whole adult schistosomes, less present in tegumental membranes and far less present in blood cells (Table 6.1), is plotted at with a high loading on both discriminant 1, representing the differences between blood cells and schistosome lipids (Fig. S1B, X-axis, far left), as well as with a high loading on discriminant 2, representing the differences between the PS species in tegumental membranes and those of whole adult worms (Fig. S1B, Y-axis, far to the bottom).
| Whole worm (n=5) |
Tegument (n=3) |
Hamster blood cells (n=5) |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| m/z | Acyls | % | ± | S.D. | % | ± | S.D. | % | ± | S.D. | |
| 718 | 16:0/18:1 | 2.8 | ± | 0.3 | 5.3 | ± | 0.5 | 7.5 | ± | 0.5 | |
| 740 | 16:0/20:4 | 1.2 | ± | 0.2 | 1.5 | ± | 0.2 | 6.4 | ± | 0.2 | |
| 742 | 18:1/18:2 | 2.1 | ± | 0.3 | 2.5 | ± | 0.2 | 7.4 | ± | 0.4 | |
| 744 | 18:1/18:1 | 7.1 | ± | 0.6 | 8.4 | ± | 0.3 | 6.6 | ± | 0.2 | |
| 746 | 18:0/18:1 | 8.2 | ± | 0.3 | 9.5 | ± | 1.0 | 3.9 | ± | 0.2 | |
| 764 | 18:2/20:4 | 1.2 | ± | 0.1 | 1.1 | ± | 0.2 | 6.4 | ± | 0.1 | |
| 766 | 18:1/20:4 | 2.8 | ± | 0.1 | 2.9 | ± | 0.2 | 10.1 | ± | 0.1 | |
| 768 | 16:0/22:4 | 4.6 | ± | 0.2 | 6.1 | ± | 0.8 | 3.3 | ± | 0.1 | |
| 768 | 18:0/20:4 | 4.1 | ± | 0.2 | 2.3 | ± | 0.3 | 4.8 | ± | 0.1 | |
| 770 | 18:2/20:1 | 5.5 | ± | 0.1 | 6.4 | ± | 0.4 | 1.8 | ± | 0.1 | |
| 772 | 18:1/20:1 | 3.9 | ± | 0.1 | 5.5 | ± | 0.4 | * | |||
| 788 | 20:4/20:4? | * | * | 5.1 | ± | 0.3 | |||||
| 790 | 18:1/22:6? | 2.7 | ± | 0.1 | 2.0 | ± | 0.0 | 6.6 | ± | 0.2 | |
| 792 | 18:1/22:4 | 4.8 | ± | 0.4 | 3.1 | ± | 0.5 | 4.0 | ± | 0.1 | |
| 794 | 18:0/22:5 | 6.3 | ± | 0.5 | 5.5 | ± | 0.8 | 3.8 | ± | 0.3 | |
| 796 | 18:0/22:4 | 17.9 | ± | 1.0 | 15.0 | ± | 2.1 | 1.2 | ± | 0.1 | |
| 798 | 18:0/20:3 | 4.3 | ± | 0.4 | 4.7 | ± | 0.3 | * | |||
| 818 | 20:1/22:6 | 4.5 | ± | 0.5 | 3.2 | ± | 0.2 | * | |||
| Whole worm (n=5) |
Tegument (n=3) |
Hamster blood cells (n=5) |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| m/z | Acyls | % | ± | S.D. | % | ± | S.D. | % | ± | S.D. | |
| 720 | 16:0/16:0 alk | 1.5 | ± | 0.1 | 5.8 | ± | 1.3 | * | |||
| 734 | 16:0/16:0 | 11.5 | ± | 0.7 | 23.1 | ± | 2.2 | 6.3 | ± | 0.2 | |
| 746 | 34:1 alk | 1.8 | ± | 0.1 | 2.2 | ± | 0.3 | * | |||
| 758 | 16:0/18:2 | 4.8 | ± | 0.4 | 5.1 | ± | 0.2 | 19.8 | ± | 1.1 | |
| 760 | 16:0/18:1 Δ9 | 11.7 | ± | 0.6 | 8.2 | ± | 0.2 | 14.4 | ± | 0.7 | |
| 760 | 16:0/18:1 Δ5 | 2.2 | ± | 0.1 | 12.4 | ± | 0.3 | * | |||
| 762 | 16:0/18:0 | 4.6 | ± | 0.2 | 3.9 | ± | 0.1 | 2.5 | ± | 0.1 | |
| 782 | 16:0/20:4 | 3.5 | ± | 0.4 | 2.2 | ± | 0.7 | 6.6 | ± | 0.3 | |
| 784 | 18:1/18:2 | 2.4 | ± | 0.1 | 2.3 | ± | 0.1 | 3.8 | ± | 0.2 | |
| 786 | 18:0/18:2 | 10.1 | ± | 0.3 | 7.0 | ± | 0.5 | 11.9 | ± | 0.6 | |
| 788 | 16:0/20:1 | 13.8 | ± | 0.4 | 5.7 | ± | 0.5 | * | |||
| 788 | 18:0/18:1 | * | * | 3.7 | ± | 0.3 | |||||
| 806 | 16:0/22:6 | 2.0 | ± | 0.2 | 1.4 | ± | 0.3 | 6.9 | ± | 0.6 | |
| 808 | 18:1/20:4 | 1.5 | ± | 0.1 | * | 3.0 | ± | 0.2 | |||
| 810 | 18:0/20:4 | 2.4 | ± | 0.2 | 1.3 | ± | 0.2 | 4.1 | ± | 0.3 | |
| Whole worm (n=5) |
Tegument (n=3) |
Hamster blood cells (n=5) |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| m/z | Acyls | % | ± | S.D. | % | ± | S.D. | % | ± | S.D. | |
| 833 | 16:0/18:2 | 4.0 | ± | 1.6 | 2.2 | ± | 0.2 | 7.1 | ± | 0.7 | |
| 835 | 16:0/18:1 | 3.1 | ± | 0.6 | 3.7 | ± | 0.6 | 2.4 | ± | 0.4 | |
| 837 | 16:0/18:0 | 2.3 | ± | 0.6 | 6.3 | ± | 0.9 | * | |||
| 857 | 16:0/20:4 | 1.1 | ± | 0.1 | * | 6.6 | ± | 0.6 | |||
| 859 | 18:1/18:2 | 1.3 | ± | 0.0 | 1.2 | ± | 0.2 | 3.4 | ± | 0.2 | |
| 861 | 18:0/18:2 | 15.7 | ± | 1.3 | 13.2 | ± | 1.3 | 13.0 | ± | 0.7 | |
| 863 | 18:0/18:1 | 21.0 | ± | 2.5 | 27.3 | ± | 1.9 | 2.3 | ± | 0.1 | |
| 865 | 18:0/18:0 | 2.3 | ± | 0.3 | 2.7 | ± | 0.1 | * | |||
| 883 | 18:1/20:4 | * | * | 5.2 | ± | 0.1 | |||||
| 885 | 18:0/20:4 | 25.0 | ± | 1.9 | 9.9 | ± | 0.8 | 36.8 | ± | 2.1 | |
| 887 | 18:0/20:3 | 3.4 | ± | 0.4 | 3.9 | ± | 0.6 | 5.7 | ± | 0.3 | |
| 889 | 38:2 | 2.2 | ± | 0.2 | 4.3 | ± | 0.6 | 1.2 | ± | 0.2 | |
| 891 | 18:0/20:1 | 1.4 | ± | 0.1 | 3.3 | ± | 0.5 | * | |||
| 911 | 40:5 | * | * | 2.8 | ± | 0.3 | |||||
| 945 | 42:2 | * | 2.3 | ± | 0.2 | * | |||||