Chapter 5
Evaluation of molecular and serological methods for diagnosing two cases of neuroschistosomiasis
Affiliations
- E1
- Department of Medical Microbiology and Infectious Diseases, Erasmus MC, Rotterdam, The Netherlands
- E2
- Laboratory of Parasitology, Harbour Hospital and Institute for Tropical Diseases, Rotterdam, The Netherlands
Abstract:
Neuroschistosomiasis is the most severe ectopic form of schistosomiasis. To prevent severe neurological damage and permanent disability, rapid diagnosis and treatment are crucial. However, diagnostic tools for rapid diagnosis of neuroschistosomiasis are currently lacking. Here we present two cases of confirmed schistosome myelopathy and compared molecular as well as distinct serological methods for their usefulness to diagnose neuroschistosomiasis. Real-time quantitative PCR and Western blots did not provide positive test results for our patients, but quantification of specific antibody levels against adult worm antigens and egg antigens in serum and cerebrospinal fluid was indicative for intrathecal antibody production directed against schistosome eggs. Hence the proposed egg/worm antibody index can be a valuable tool to distinguish between on the one hand patients with true neuroschistosomiasis and on the other hand patients that have systemic schistosomiasis but no eggs in the spinal cord, where the transverse myelitis is caused by a different disorder.
Introduction
Schistosomiasis is a tropical disease affecting an estimated 200 million people worldwide, mainly in sub-Saharan Africa (74). The disease is caused by a parasitic flatworm of the Schistosoma genus. Eggs laid by the female parasite that are not excreted accumulate in host tissue and cause the vast majority of the disease symptoms. Adult worms usually reside in the mesenteric veins or in the blood vessels surrounding the urine bladder. Sometimes, however, worm pairs reside in ectopic locations. The most severe ectopic form of schistosomiasis is transverse myelitis. Transverse myelitis is an inflammation of the spinal cord causing neurological dysfunction such as muscle weakness, sensory loss, bladder dysfunction and finally paraplegia. In case of neuroschistosomiasis, it is caused by the inflammatory response of the host to the schistosome eggs that are trapped in the spinal cord. The eggs are deposited in situ after aberrant migration of adult worms to the central nervous system (CNS), or by egg dissemination via the bloodstream through retrograde venous flow into the Batson vertebral epidural plexus, which connects the deep iliac veins and inferior vena cava with veins of the spinal cord (26,27,154). The inflammatory lesions around the eggs suppress the spinal cord causing neurological dysfunction. Neuroschistosomiasis has an acute or subacute onset and may then progress to full paralysis within 15 days if untreated (63). Diagnosis of neuroschistosomiasis is difficult and the disorder is often not recognized and therefore probably underdiagnosed (62). True prevalence is unknown, but a study in Brazil showed that 5.6% of non-traumatic and non-tumoural myelopathies were caused by neuroschistosomiasis (29).
Normally, the cerebrospinal fluid (CSF) contains little protein and few cells. During inflammatory diseases of the CNS, the blood-brain-barrier is usually damaged at least to some extent. Therefore, leakage of antibodies from blood plasma into the CSF is likely to occur. Hence, the presence of anti-schistosome antibodies in the CSF could either be the result of intrathecal antibody production due to neuroschistosomiasis or it could be the result of leakage of antibodies from the plasma into the CSF due to damage to the blood-brain-barrier caused by a distinct neurological disorder. Leakage of plasma antibodies into the CSF is a-specific and the profile of the antibodies leaking will resemble that of plasma antibodies. Intrathecal antibody production on the other hand is specific and the antibodies produced are directed against the antigens of the pathogen in the central nervous system. As the number of (migrated) B-cells in the CSF is limited, the different antibodies that are produced intrathecally are also limited. In case of neuroschistosomiasis, where the immune response is directed against the schistosome eggs that have crossed the blood brain barrier, intrathecal antibody production is expected to be differentially directed to eggs more than to worms. However, it should be realized that the egg and the adult worm share many of their antigenic proteins (40).
As therapeutic delay may quickly lead to severe neurological damage and permanent disability, rapid diagnostic tools are needed (28). Diagnosis of spinal cord neuroschistosomiasis usually relies on the clinical diagnosis of transverse myelitis by imaging techniques (such as MRI) to show edema and swelling of the spinal cord, in combination with diagnosis of an active infection with Schistosoma spp. and exclusion of other causes of myelitis. Current methods cannot properly discriminate between neuroschistosomiasis as the cause of the transverse myelitis and transverse myelitis with other causes in patients with systemic schistosomiasis. The gold standard for diagnosis of neuroschistosomiasis is retrieval of eggs from the spinal cord (28). However, this invasive method is rarely used, because of the risk for serious complications. Therefore, all current methods of diagnosis are non-specific for neuroschistosomiasis and are based on exclusion of other causes of the myelopathy. It is important to exclude any other infectious cause of myelopathy and to be certain of the diagnosis neuroschistosomiasis because the therapy for neuroschistosomiasis includes a high dose of corticosteroids that will have adverse effects in patients with transverse myelitis caused by viral or bacterial pathogens (183).
Here we report two cases of schistosome myelopathy and examined the usefulness of three methods to diagnose neuroschistosomiasis. These methods have proven to be useful in the diagnosis of other infections in the CNS such as neurosyphilis, toxoplasmosis, and varicella zoster virus infection. The following tests were examined: (1) detection of schistosome DNA in CSF by real-time semi-quantitative PCR in order to demonstrate the presence of the parasite in the CNS, (2) Western blot analysis to demonstrate a qualitatively different anti-schistosome antibody composition in CSF compared to serum, and (3) indirect hemagglutination assay (IHA) and ELISA to demonstrate a quantitatively different anti-schistosome antibody composition in CSF compared to serum, a method previously described (42).
Materials and Methods
Case descriptions
Two patients (A and B) presented at the Erasmus University Medical Center with the clinical symptoms of progressive transverse myelitis. Paired serum and CSF samples from each patient were taken at presentation and once thereafter.
Patient A was a 35 year-old male who in May 2008 presented to the emergency department with progressive pain and muscle weakness in the upper legs (42). Two months prior to presentation, he suffered from diarrhea during a visit to Brazil. The pain in his legs started four days prior to hospital presentation and was accompanied by dysuria and a feeling of incomplete voiding. During the next few days, muscle weakness, sensory disturbances and bladder dysfunction increased. An MRI scan showed swelling and edema of the low-thoracic and lumbar spinal cord without abnormalities in the brain. Antibodies against schistosome antigens were found in serum and CSF and levels were increased after 11 days (Table 5.1). In a rectal biopsy one dead S. mansoni egg embedded in a granuloma was found. Based on these results and the negative test results for other infectious causes of transverse myelitis, the diagnosis neuroschistosomiasis was made and treatment was started. The patient received praziquantel (2 daily doses of 30 mg/kg for 3 days) and high dose of intravenous (i.v.) methylprednisolone (2 daily doses of 500 mg for 5 days) followed by oral prednisone (3 daily doses of 0.5 mg/kg) tapered gradually over the next several months. Within a week the patient's condition improved dramatically and he was discharged from the hospital. During the next months, muscle strength and sensibility improved, and bladder function normalized.
Patient B was a 59-year old male who presented in September 2010 at our neurology department. The patient was born in Suriname, a country to which he traveled regularly and his last visit was in 2009. In January 2010 he developed progressive lumbar pain irradiating to the lower limbs, with asymmetric loss of sensibility and progressive muscle weakness in the legs. The patient was able to walk for 15 to 30 minutes, but fell regularly. He had problems walking stairs and needed to pull himself on the handrail. The patient was incontinent for urine when in high need. Otherwise miction and defecation were not affected. MRI showed a lesion in the spinal cord at the low-thoracic and lumbar typical for myelitis transversa. The protein level (1.12 g/L) and the numbers of leukocytes (19×106/L) and monocytes (13×106/L) were elevated in CSF. Furthermore, antibodies against schistosomes were found in paired serum and CSF samples, collected at presentation and 8 days later (Table 5.1). No eggs of schistosomes could be demonstrated by microscopic methods in feces or urine. Since no other cause for the transverse myelitis was found, the patient was treated with praziquantel (2 daily doses of 30 mg/kg for 3 days) and methylprednisolone (once daily 500 mg i.v. for 5 days) followed by oral prednisone (2 daily doses of 0.5 mg/kg tapered gradually over the next several months). The physical condition of this chronic patient improved significantly within a week, but the patient did not fully recover probably due to irreversible nerve damage caused in preceding nine months of disease.
DNA isolation and real-time PCR
A real-time PCR was performed on DNA extracted from 0.2 ml CSF using easyMag (bioMérieux, France) according to the manufacturer's instructions. Phocine Herpes Virus (PhHV) was added to the lysis buffer as an internal control. The used primers and probes have been designed based on sequences from S. haematobium (GenBank accession DQ677661), S. mansoni (AF503487) and S. intercalatum (U22166) (127). We used the following PCR cycle conditions: a denaturation step of 15 min at 95 °C, followed by 50 cycles of 15s denaturation at 95 °C, 30s annealing at 60 °C and 30s elongation at 72 °C. Fluorescence was measured at each annealing step.
Western blot analysis
Worms and eggs were retrieved from experimentally infected hamsters. Worms and eggs were homogenized by sonification for 3 30 sec in phosphate buffered saline (PBS). The homogenate was spun down for 10 min at 18000 g. The obtained supernatant was used to load SDS-PAGE gels (5 μg adult worm protein or 10 μg egg protein, as measured by a Lowry protein assay using bovine serum albumin as a standard). Gels were blotted on PVDF membranes, blocked for 1 hr in low fat milk powder in TBS-tween and subsequently incubated with either serum (1:1000) or CSF (1:100). The secondary antibody was HRP conjugated anti-human IgG (1:10000) (Dako, Glostrup, Denmark). Bands were visualized using ECL (Pierce, Rockford, IL, USA) on Hyperfilm (Amersham, GE healthcare, Diegem, Belgium).
Serological methods
Total albumin and total IgG were determined in serum and CSF by routine clinical chemistry methods. Antibody titers to schistosome worm-antigens were determined using a commercial IHA (Fumouze Diagnostics, Levallois-Perret, France) and agglutination at serum titers of 1:80 and over are positive for schistosomiasis (189). Schistosome egg-specific antibodies were determined by ELISA of soluble worm antigen (SEA) as described before (189). Anti-egg antibody levels were expressed as Arbitrary Units (AU) calculated as Optical Density (OD) dilution/10 and AU values 88 in serum are positive test results for schistosomiasis.
Theory and calculation of serological indices
The albumin index is a measure for the integrity of the blood-brain barrier and values ≥0.0090 indicate an impaired function of the blood-brain barrier (181).
- AC = albumin concentration in CSF (g/L)
- AS = albumin concentration in serum (g/L)
The total IgG index is a measure for increased concentrations of total IgG in CSF and values ≥0.70 are already suggestive for intrathecal antibody production, since albumin leaks more easily through the blood-brain barrier when compared to antibodies (104,60,142).
- IC = total IgG CSF (g/L)
- IS = total IgG serum (g/L)
Intrathecal antibody production will occur upon any inflammatory response in the CNS, and therefore, the total IgG index is not specific for schistosomiasis. Demonstration of schistosome-specific antibodies in CSF is thus more informative and this has indeed been shown to be an indication for neuroschistosomiasis (61,131). However, the presence of schistosomes-specific antibodies in CSF does not discriminate intrathecal production of these antibodies from antibody leakage from the plasma into the CSF. Two methods have been developed to correct for leakage of antibodies from plasma into the CSF. Antibody titers can be corrected for albumin concentrations in CSF, which is not commonly used, or corrected for total IgG in CSF, which is known as the specific antibody index. The cut-off value for specific antibody indices is 1.5, with higher levels indicative for intrathecal production of specific antibodies (143).
- EC = anti-egg IgG in CSF (OD × dilution)
- ES = anti-egg IgG in serum (OD × dilution)
Transverse myelitis in neuroschistosomiasis is caused by the immune response directed to schistosome eggs. To differentiate between leakage of plasma antibodies and intrathecal antibody production, we also used an index which calculates whether the anti-schistosome IgG response in CSF is more egg-directed than in serum. We used the egg-worm antibody index as a measure for the relative enrichment of anti-egg IgG over anti-worm IgG in CSF when compared to serum.
- WC = anti-worm IgG in CSF (titer)
- WS = anti-worm IgG in serum (titer)
This is the most specific antibody index which will only show increased values when antibodies directed against eggs of schistosomes are produced intrathecally, which only occurs when neuroschistosomiasis is the cause of the transverse myelitis. Following Luger et al. (2000)(104), we suggest a cut-off value of 2 for this egg-worm antibody index, representing a two-fold increase in egg-specific antibodies in CSF. This means that values ≥2 indicate intrathecal production of IgG specific for schistosome eggs, and thus indicate neuroschistosomiasis.
| Sample | Type | Albumin (g/L) |
Total IgG (g/L) |
Worm IHA titer |
SEA ELISA AU |
|---|---|---|---|---|---|
| A1 | serum | 43.3 | 12.4 | 2560 | 229.6 |
| CSF | 0.859 | 0.128 | 16 | 3.95 | |
| A11 | serum | 41.2 | 9.8 | 5120 | 286.8 |
| CSF | 0.771 | 0.228 | 32 | 7.73 | |
| B1 | serum | 44.0 | 13.3 | 5120 | 136.0 |
| CSF | 0.803 | 0.169 | 4 | 1.88 | |
| B8 | serum | 46.7 | 13.7 | 5120 | 185.2 |
| CSF | o.438 | 0.070 | 4 | 1.63 |
Results
Real-time semi-quantitative PCR
Real-time semi-quantitative PCR is a very sensitive method to detect specific DNA and it has been used to detect schistosome infections in blood (36,198,204). We hypothesized that if the transverse myelitis was caused by eggs of Schistosoma species, some of their DNA might leak into the CSF and this schistosome DNA can then be detected by a sensitive PCR method. Hence, for our two patient cases we examined the usefulness of real-time semi-quantitative PCR on CSF for the diagnosis of neuroschistosomiasis. The used PCR method targets the internal-transcribed-spacer-2 (ITS2), a multi-copy sequence in schistosomes. This assay had a detection limit of 0.1 pg of schistosome DNA per assay as determined in our laboratory by the use of dilution series of purified chromosomal DNA of adult schistosomes (not shown). Thus, the detection limit is less than the amount of DNA present in one single schistosome cell. Despite the high sensitivity of the PCR, schistosome DNA could not be amplified in any of the CSF samples of our two neuroschistosomiasis patients while internal controls were amplified properly. Therefore, detection of schistosomal DNA by a sensitive PCR method does not seem to be useful for diagnosing neuroschistosomiasis.
Serology
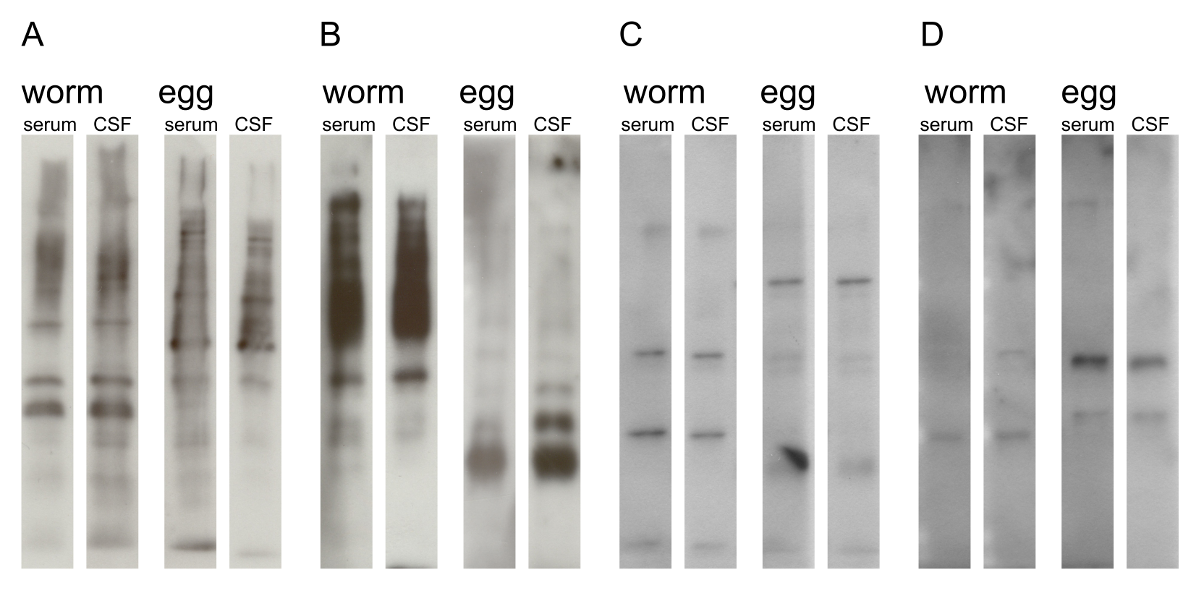
When intrathecal antibody production against schistosome antigens can be demonstrated, neuroschistosomiasis is confirmed. Therefore, it is important to distinguish between leakage of antibodies from blood plasma into the CSF and intrathecal antibody production. In the case of leakage of antibodies against schistosomes from the plasma into the CSF, the cause of the transverse myelitis is not necessarily neuroschistosomiasis but could be anything that causes damage to the blood-brain-barrier in patients that are coincidently infected with Schistosoma species. Intrathecal antibody production is characterized by a raised total IgG index and/or oligoclonal bands in the CSF. Serological diagnosis of neuroschistosomiasis can only be made when intrathecal production of specific antibodies against schistosomes can be demonstrated. We performed Western blots, IHA and ELISA to qualify and quantify the anti-schistosome response in both serum and CSF of two patients with neuroschistosomiasis to examine their usefulness for the diagnosis of neuroschistosomiasis. Western blots using worm and egg fractions were performed with both serum and CSF to detect intrathecal production of schistosome-specific antibody production as prove for neuroschistosomiasis (Fig. 5.1). Many protein bands were detected by Western blot using serum and CSF of neuroschistosomiasis patients (Fig. 5.1A and B). However, protein bands were also detected when blots were incubated with serum and CSF from patients infected with other pathogens (Fig. 5.1C and 1D), demonstrating that not all reacting protein bands are the result of schistosomes-specific antibodies. Furthermore, the banding pattern to worm and egg fractions was similar in CSF and serum from the same neuroschistosomiasis patient (Fig. 5.1A and B). In CSF of patient B the Western blot with egg antigens, shows a band at 35 kD that seems to be absent in serum, but interpretation is difficult due to the reduced staining reactivity of the serum blot compared to the CSF blot (Fig. 5.1B). Therefore, the Western blots could not reveal an obvious differential schistosome-specific antibody response in CSF versus serum in our neuroschistosomiasis patients. Hence, no schistosome-specific intrathecal antibody production could be demonstrated. These results are consistent with the findings of Pammenter et al. (1991)(131), and therefore, our results support their conclusion that there is no additional diagnostic value for Western blot over conventional ELISA in the diagnosis of neuroschistosomiasis.
We used an anti-egg ELISA and a worm IHA to quantify the antibody responses in both CSF and serum to native schistosome antigens. Albumin and total IgG were also measured (Table 1). With these measurements we were able to calculate different indices as mentioned above which are informative about the amount of damage to the blood-brain barrier, the amount of intrathecal antibody production and whether the intrathecal IgG response directed against schistosomes is egg-skewed or adult worm-skewed. In all our samples the albumin index was elevated, indicating a damaged blood brain barrier (Table 5.2). This further supports the clinical picture of transverse myelitis but does not give any clues towards the cause of the transverse myelitis. Where the albumin index is a measure for blood-brain damage, the total IgG index can indicate intrathecal antibody production. An elevated total IgG index in schistosome-infected patients is indicative for neuroschistosomiasis (60), but is not specific for neuroschistosomiasis and may occur in any infectious cause of transverse myelopathy. Only one of each of our paired serum/CSF samples of the two patients had an increased total IgG index indicating an infectious cause of the transverse myelitis (Table 5.2). Hence, this index does not seem to be a sensitive method for the diagnosis of transverse myelitis caused by infectious agents.
Pammenter et al. (1991)(131) and Ferrari et al. (1995)(61) showed that the presence of schistosome-specific IgG in CSF is indicative for neuroschistosomiasis. However, this test has a low negative predictive value (131) or lacks sensitivity at the suggested cut off of 1.4 μg/ml (61). Furthermore, this test does not discriminate between antibody leakage form plasma and intrathecal antibody production. A schistosome-specific antibody index or the egg-worm antibody index can confirm intrathecal egg-specific antibody production more specifically than a total IgG index or the absolute levels of anti-worm or anti-egg IgG in the CSF. Magalhães-Santos et al. (2003)(107) noted that the IgG1 isotype levels were higher in the CSF than in serum of patients with neuroschistosomiasis. This suggests local synthesis of IgG1. However, they did not calculate the specific IgG1 antibody index to test for specific intrathecal antibody production. Two of our four samples tested had a positive schistosome-specific antibody index - one from each patient - showing that this antibody index is more sensitive than the total IgG index, but still not sensitive enough for the diagnosis of neuroschistosomiasis. Thus, in our patients, the schistosome-specific antibody index (not divided by subclasses) was not sensitive enough to diagnose neuroschistosomiasis as the cause of transverse myelitis.
However, the egg-worm antibody index did indicate intrathecal antibody production in all paired serum-CSF samples (Table 5.2), thereby confirming neuroschistosomiasis. Both our patients had increased indices. Patient A had a slightly positive index for the first paired serum/CSF sample which had increased to 4.31 after 11 days. Patient B had highly elevated indices at both time points.
| Sample | Albumin index |
Total IgG index |
Schistosome specific antibody index |
Egg-worm antibody index |
|---|---|---|---|---|
| A0 | 0.020 | 0.52 | 1.7 | 2.75 |
| A11 | 0.019 | 1.24 | 1.2 | 4.31 |
| B0 | 0.018 | 0.70 | 1.1 | 17.65 |
| B8 | 0.009 | 0.55 | 1.7 | 11.27 |
Discussion
Although PCR detection of viral and bacterial pathogens can be used to diagnose several infectious diseases in the CNS, this technique was not capable of detecting schistosome DNA in the CSF of confirmed neuroschistosomiasis patients. Apparently, insufficient amounts of DNA of the eggs are present in the CSF. Viral and bacterial pathogens infecting the CNS replicate and in general disseminate, thereby multiplying their amount of DNA in spreading areas of the CNS. These living micro-organisms or DNA fragments of them can thus be retrieved in the CSF sample collected by puncture. However, schistosomes nor their eggs multiply within the host. Thus, the amount of schistosome DNA from the eggs in the CSF is relatively restricted. Furthermore, the eggs will be encapsulated in granulomas formed by the inflammatory immune response that is elicited upon protein excretion by the developing miracidium inside the egg, prohibiting extensive diffusion of DNA into the CSF. We conclude that real-time semi-quantitative PCR has no additional value in the diagnosis of neuroschistosomiasis, although PCR on stool samples can be useful for confirmation of an active schistosome infection in addition to serology and traditional microscopy (180).
In addition to DNA amplification methods, we examined various serological methods for the usefulness to diagnose neuroschistosomiasis. Intrathecal antibody production against schistosomal eggs could not be demonstrated by Western blot techniques, but the schistosome-specific antibody index and especially the egg-worm antibody index were indicative for neuroschistosomiasis in our patient cases. A further advantage of the egg-worm antibody index is that the outcome is independent of the diagnostic method used. It can be calculated with the results of any two serological tests for the diagnosis of schistosomiasis based on egg and worm antigens. Therefore, this concept could also be useful to diagnose neuroschistosomiasis caused by S. japonicum and S. mekongi, the schistosome species present in Asia, by using serological methods detecting egg and worm antigens of these species (99,101). Every laboratory can implement a ratio with the diagnostic methods that are currently in use in that laboratory. In addition, the test is not biased by the presence of cross reacting antibodies in serum from previous infections, as a positive egg-worm antibody index can only be achieved by increased levels of antibodies in CSF due to intrathecal production. On the other hand, all four test results need to be positive in order to calculate the index, which might limit the sensitivity of this test in endemic areas as antibody titers against schistosomes tend to decrease in time during chronic schistosomiasis due to immune suppression induced by the schistosomes (193).
In conclusion, quantification of antibody levels against worm and egg antigens in CSF and serum to determine the egg-worm antibody index is a simple and relatively rapid method that seems to be useful in the diagnosis of neuroschistosomiasis. However, confirmation on a larger set of neuroschistosomiasis patients is required for the ultimate validation of this method.