Chapter 4
Binding of von Willebrand factor and plasma proteins to the eggshell of Schistosoma mansoni
Affiliations
- D1
- Department of Medical Microbiology and Infectious Diseases, Erasmus MC, Rotterdam, The Netherlands
- D2
- Center for Proteomics and Metabolomics, Leiden University Medical Center, Leiden, The Netherlands
- D3
- Department of Parasitology, Center for Infectious Diseases, Leiden University Medical Center, Leiden, The Netherlands
- D4
- Department of Clinical Chemistry and Haematology, University Medical Center Utrecht, The Netherlands
Abstract:
Schistosoma mansoni eggs have to cross the endothelium and intestinal wall to leave the host and continue the life cycle. Mechanisms involved in this essential step are largely unknown. Here we describe direct binding to the S. mansoni eggshell of von Willebrand factor and other plasma proteins involved in haemostasis. Using deletion-mutants, we demonstrated that it is the A1 domain of von Willebrand factor that binds to the eggshell. Our results suggest that binding of plasma proteins to the eggshell promotes binding to the endothelium, initiating the passage of the egg through the blood-vessel wall to be excreted in the end.
Main article
Schistosoma spp. are helminths causing the tropical disease schistosomiasis. An estimated 200 million people are affected, most of them living in sub-Saharan Africa (74). These parasites penetrate their human host through the skin after which the cercariae transform into schistosomula that subsequently enter the circulation. In the case of Schistosoma mansoni, male and female worms meet in the portal vein, where they pair and mature before moving to the mesenteric veins, their final habitat (74). Mature female worms excrete approximately 300 eggs per day, which is about one every 5 minutes (32). In order to complete the schistosome life-cycle, the eggs have to be excreted with the faeces. Therefore, eggs have to cross the blood-vessel wall as well as the intestinal wall. Many eggs fail to extravasate and are transported by the blood flow to the liver where they get stuck and induce granuloma formation, fibrosis of the liver and portal hypertension. These non-excreted eggs are the major cause of morbidity in schistosomiasis.
Eggs have a rigid shell structure of cross-linked proteins and thus cannot propel themselves (48). Therefore, eggs depend on external forces in order to cross the host tissues. Mechanisms involved in excretion of eggs are largely unknown, even though this is an essential step in the life-cycle of schistosomes. Eggs of several schistosome species have a sharp spine and it could be imagined that the presence of this spine aids in passing through the obstructing structures of the host (89). However, eggs of Schistosoma japonicum and Schistosoma mekongi lack a sharp spine, which makes this theory unrealistic. It is known that the process of excretion is partly dependent on the host immune reaction directed towards eggs (49), but additional mechanisms are likely to be involved.
It has been demonstrated that eggs attach to endothelium and that endothelial cells actively migrate over the eggs (64). The endothelial surface-adhesion molecules, ICAM-1, E-selectin and VCAM-1, and surface exposed Lewis X glycans on S. mansoni eggs play an important role in egg adhesion to the vascular endothelium (98). In addition, plasma factors enhance egg attachment to endothelial cells in vitro and platelets have a role in extravasation and excretion in vivo (64,125,126). Furthermore, schistosome eggs were also shown to be a potent and direct activator of platelets and platelets were shown to adhere en masse to eggs in vitro (125,203). Activated platelets aggregate and stimulate secondary coagulation (the plasma clotting factor cascade) which results in fibrin fibre formation to stabilise the platelet plug. Apparently, there is an interaction of the schistosome eggshell with the host haemostatic system, but the molecular structures involved in the activation of the haemostatic system are unknown.
We hypothesised that the egg probably attracts host proteins and host cells in order to hold onto the vessel wall and help passage through the endothelium and the gut wall, allowing excretion from the host and continuation of the life cycle. Therefore, we analysed the interaction of S. mansoni eggshells with von Willebrand factor (VWF), as this clotting factor is crucial for anchoring clotting material to damaged or activated surfaces of the blood vessel, and VWF could thus play an essential role in egg extravasation. VWF is an adhesive glycoprotein of 250 kD which forms multimers that can be extremely large and consist of over 80 subunits, resulting in a multimer of over 20 mega-Dalton. VWF contains many binding domains that can connect platelets to clotting factors and injured surfaces of the endothelium (105,159). VWF binds platelet glycoprotein Ib (GPIb) on platelets which are thereby activated and initiate clot formation.
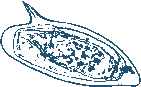
The presence of VWF on S. mansoni eggs isolated from collagenase digested livers of infected hamsters was demonstrated by immunofluorescence (Fig. 4.1A). As these eggs were directly obtained from perfused livers, this demonstrates that VWF binding occurred in vivo in the blood vessel.
Schistosome eggs were also incubated in vitro with purified human VWF. We used both untreated eggs and stripped eggs. From stripped eggs, all non-covalently bound proteins were removed by a procedure which includes multiple washes with high salt conditions, detergents, urea and even boiling in SDS, in order to be entirely sure that no host factors remained on the surface of the schistosome egg (47). Both untreated (Fig. 4.1C) and stripped eggs (Fig. 4.1D) bound purified human VWF in vitro, demonstrating the direct interaction between VWF and the eggshell. The binding of VWF to eggs could also be demonstrated at a more quantitative level using stripped eggshell fragments and enhanced chemiluminescence (ECL) measurements (Fig. 4.1H).
We also incubated stripped schistosome eggs for 1 min with citrate plasma from healthy adult human volunteers, obtained after informed consent. Binding of VWF to eggs was then demonstrated by immune fluorescence (Fig. 4.1F and G). This result demonstrated that VWF in the presence of all other plasma proteins still binds to the schistosome eggs, although the punctuated fluorescence pattern suggests that other plasma proteins compete for egg binding, or that other plasma proteins shield bound VWF prohibiting its detection by immune fluorescence. Altogether these results demonstrate that there is a direct interaction between schistosome eggshells and VWF. To determine which VWF domain is involved in eggshell binding, we incubated eggs with recombinant human VWF of which either the A1 (97), A2 (95) or A3 domain (96) was deleted. Reduced VWF binding was only observed with the A1 domain-deleted VWF, but not with VWF lacking the A2 or A3 domain (Fig. 4.1I). This indicates that VWF binds eggshells with its A1 domain. Domain A1 of VWF is a relatively small part of VWF as it comprises circa 250 amino acids, which is approximately 15% of the entire VWF protein (97). In addition, deletion of the A1 domain does not affect the structure and function of the other domains in VWF (95,96,97). Hence, the dependence on a small domain of VWF for its binding to schistosome eggs suggests a specific interaction.
The A1 domain of VWF also binds GPIb, a glycosylated receptor on platelets. Since the eggshell contains glycan structures as well (47), we hypothesised that eggshell glycans may play a role in the binding of VWF. However, we could not find indications for glycan-mediated VWF binding to eggshells. Treatment of eggshells with glycosidases did not reduce VWF binding (data not shown), but these treatments did not remove all glycan structures either and therefore these experiments were inconclusive. Blocking of glycan access with monoclonal antibodies against schistosome glycans (291-2G3-A, 114-3A5, 128-4F9-A (which bind to Galβ1-4(Fucα1-3)GlcNAc (Lewis X)), 273-3F2 (which binds to GalNAcβ1-4GlcNAc (LDN)) (192) and 114-4D12 (which binds to Fucα1-2Fucα1-3 (DF) termini) (148)) had no effect on the binding of VWF to eggshells (data not shown). Furthermore, treatment of eggshells with periodate, which disrupts glycan structures, did not reduce VWF binding (data not shown). In conclusion, VWF binds directly to eggshells through its A1 domain, most likely in a non-glycan-mediated manner.
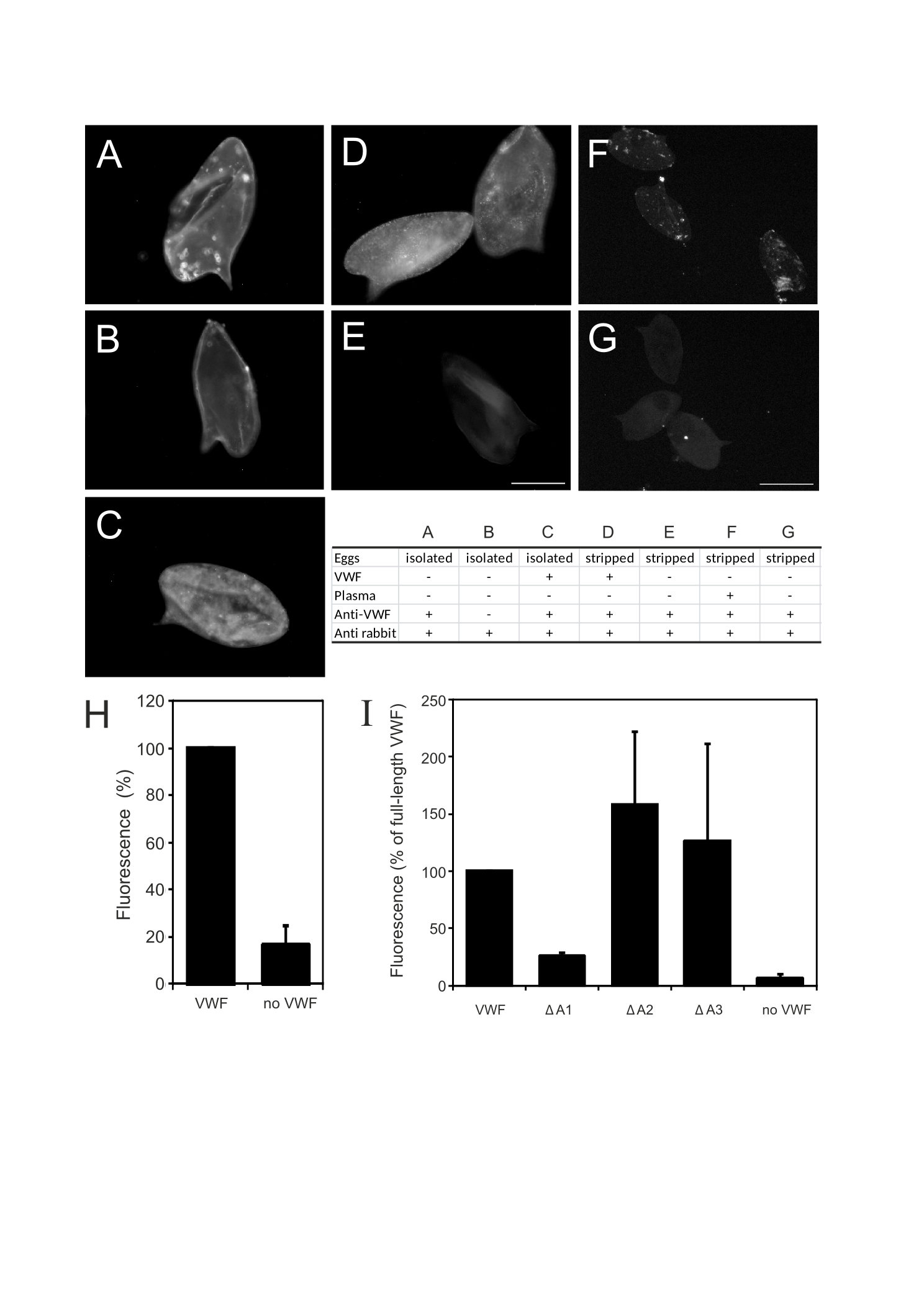
When eggs are laid by the female schistosome in the circulation of the host, they immediately come into contact with circulating plasma proteins. We hypothesised that, next to VWF, other plasma proteins involved in fibrin formation, platelet activation and aggregation, bind to eggshells. To test this, we incubated stripped eggshell fragments with plasma and subsequently washed those six times with PBS. Thereafter, bound plasma proteins were released by boiling the eggshells in Laemmli buffer. Released proteins were analysed by SDS-PAGE together with a lane of diluted plasma (Fig. 4.2). A clear difference in protein band pattern was observed between plasma and plasma proteins bound to schistosome eggshell fragments, especially in the high molecular weight range, demonstrating selective binding of certain plasma proteins to eggshells. Following trypsin digestion and LC-MS/MS analysis, the identified proteins in both samples were compared (Table 4.1 and Supplementary Table S1). The high molecular weight plasma protein which specifically bound to eggshells (gel slices 1 and 2) was identified as fibronectin. Although the pattern of protein bands between approx. 50-75 kDa in plasma was similar to the pattern of proteins bound to eggshells, the major proteins in these gel slices were different. In this respect it is noteworthy that albumin, the major plasma protein, could hardly be detected among the proteins bound to eggshells. This validates the selectivity of the binding. The plasma proteins specifically bound to eggshells in the molecular weight region of albumin consisted mainly of fibrinogen (gel slices 5-7). Fibrinogen is a haemostatic protein and is the precursor of fibrin. When cross-linked, fibrin networks reinforce a platelet plug. Similar to fibrin, fibronectin plays an essential role in wound healing and blood clot formation.
| Total plasma proteins | Eggshell-bound plasma proteins | |||
|---|---|---|---|---|
| Gel slice number |
Protein name | Peptides detected |
Protein name | Peptides detecteda |
| 1 | Albumin | 19 | Fibronectin 1 | 80 |
| 2 | Albumin | 22 | Fibronectin 1 | 139 |
| Fibronectin 1 | 13 | Talin 1 | 7 | |
| Alpha-2-macroglobulin | 11 | - | ||
| 3 | Albumin | 36 | Fibronectin 1 | 34 |
| Alpha-2-macroglobulin | 25 | Collagen, type II, alpha 1 | 5 | |
| Ceruloplasmin (ferroxidase) | 11 | - | ||
| 4 | - | Fibronectin 1 | 9 | |
| 5 | Albumin | 64 | - | |
| Serotransferrin | 26 | - | ||
| 6 | Albumin | 132 | Fibrinogen alpha chain | 57 |
| Fibrinogen alpha chain | 15 | Vitronectin | 14 | |
| Uncharacterised protein | 8 | Albumin | 14 | |
| 7 | Albumin | 42 | Fibrinogen beta chain | 30 |
| Alpha-1-antitrypsin | 28 | Fibrinogen alpha chain | 23 | |
| Ig gamma-1 chain C region | 14 | Uncharacterised protein | 13 | |
| 8 | - | Fibrinogen gamma chain | 32 | |
| - | Fibrinogen alpha chain | 18 | ||
| - | Actin, beta | 7 | ||
| 9 | Albumin | 19 | Actin, beta | 27 |
| - | Apolipoprotein A-IV | 12 | ||
| - | Fibronectin 1 | 12 | ||
| 10 | Albumin | 25 | Fibrinogen alpha chain | 10 |
| Haptoglobin | 16 | - | ||
| Apolipoprotein A-IV | 4 | - | ||
| 11 | Albumin | 8 | Clusterin | 15 |
| - | Apolipoprotein E | 10 | ||
| - | Fibrinogen alpha chain | 9 | ||
| 12 | Albumin | 9 | Apolipoprotein E | 34 |
| Apolipoprotein E | 6 | Fibrinogen alpha chain | 10 | |
| Fibrinogen alpha chain | 6 | Fibrinogen gamma chain | 10 | |
| 13 | Uncharacterised protein | 12 | - | |
| 14 | Albumin | 11 | - | |
| 15 | Apolipoprotein A-I | 11 | - | |
| a Average of two independent experiments. | ||||
The set of proteins bound to eggshell was also enriched in actin and talin. These cytoskeleton proteins probably originate from the remaining platelets in the platelet-poor plasma. This is in accordance with previous reports which show that platelets bind very well to eggshells (203). We also observed an enrichment of apolipoproteins E and A-IV in the set of proteins that bound to eggshells (Table 1). In plasma, Apo-IV is present on chylomicrons, whereas Apo E is present on chylomicrons and on intermediate-density lipoproteins (IDLs). Their presence in the set of proteins bound to eggshells could be a result of binding of lipoproteins to eggshells. It is known that adult worms have a low-density lipoprotein (LDL)-receptor-like protein on their tegument (158) and that the worms bind LDL (34). The binding of LDL probably helps the adult worms to mask their antigens and hence hide from the immune system. However, on eggshells no LDL-receptor-like protein is known to be present.
In contrast to the immune fluorescence experiments, the proteome analysis of bound plasma proteins did not reveal VWF as a protein bound to schistosome eggs. Fluorescence is a highly sensitive detection method compared with our proteomic workflow. Mass spectrometry detects the most abundant peptides of a trypsin digest of a protein sample, and thus this technique will only show the proteins mostly present in the analysed material. Therefore, absence of detection by this method cannot be interpreted as an indication that the protein in question is not present. Of note, the plasma concentration of VWF (approximately 10 µg/ml) is very low compared with that of fibrinogen and fibronectin (2-4 mg/ml, 0.3-0.4 mg/ml, respectively). These plasma components each bound to the eggshell and were extremely enriched from plasma compared with albumin and other plasma proteins, but the unfavorable concentration ratio of VWF to fibrinogen and fibronectin on the eggshell seems to have left VWF undetectable by proteomics. The near absence of albumin in the proteins bound to eggshells further demonstrates the specificity of the binding of other plasma proteins to S. mansoni eggshells. Moreover, the binding efficiencies and kinetics of VWF compared with the other possibly competing eggshell binding proteins identified in our proteomics experiments may also have played a role.
To check whether all eggshell bound proteins were from the host and not from the parasite itself, the mass spectrometric data were also searched against the schistosome database. The only schistosome protein found in this analysis was actin, but all identified peptides showed complete homology to human actin. As no schistosome-specific peptides derived from actin were observed, it is likely that the human plasma and not the schistosome itself is the source of the actin found. Altogether, the set of eggshell-binding plasma proteins is indicative of fibrin formation and platelet binding to eggshell.
Our results demonstrate that the schistosome eggshell binds the plasma proteins, VWF, fibrinogen and fibronectin, which play an essential role in clot formation. The main function of VWF is binding to other proteins by protein-protein interaction, for which VWF comprises many binding domains. Upon injury VWF plays an important role in platelet-vessel wall adhesion by binding both platelets (via GPIb and GPIIb-IIIa) and the extracellular matrix of the damaged endothelium exposing collagen and fibrin. Binding of VWF to the GPIIb-IIIa complex, which is exposed on activated platelets only, also plays a role in platelet cross-linking and platelet plug expansion (105). Direct binding of VWF to the eggshell by the A1 domain may thus have two effects. VWF may be directly involved in the binding of eggs to the endothelium by forming a bridge between the eggshell and the extracellular matrix. Indirectly, binding of VWF in combination with the binding of platelets (203) may induce platelet adhesion, platelet activation and secondary haemostasis, allowing the formation of a stable clot. In turn, this should facilitate a firm binding of the egg to the endothelium. Next to induction of clot formation, the schistosome egg also contains fibrinolytic capacity, as it has been shown that soluble extracts of eggs contain a plasmin-like fibrinolytic enzyme activity that could counteract uncontrolled thrombogenesis (51).
Coagulation normally occurs in the case of bleeding, when endothelium is damaged and extracellular matrix is exposed to which VWF can bind. Damage to the endothelium is in itself an activator of platelets and blood coagulation, as are shear stress and turbulence (177). In case of schistosomiasis, shear stress can be caused by the presence of the relatively large worms and eggs. Attachment of adult worms to the endothelium may cause endothelial damage that leads to activation of endothelial cells (168). As coagulation is initiated on eggshells in the near presence of damaged and activated endothelium, VWF bound to the egg may anchor the eggshell to the endothelium, after which platelet aggregation and fibrin clot formation can stabilise the binding.
Extravasation is the first step of excretion and is likely to occur rapidly, before eggs have fully matured and start secreting enzymes and other material. In vitro adhesion of eggs to endothelium takes place within 2 h (98,126). Within 4 h after adhesion to the vascular endothelium, endothelial cells actively migrate over the eggs (64). This way the eggs are removed from the blood vessel. The binding site by which VWF binds to S. mansoni eggs is not easy to resolve experimentally, as this target cannot be isolated because the eggshell is composed of a heavily cross-linked protein structure (48). Furthermore, VWF is a very large protein that has no catalytic activity and its primary function is binding to other proteins via protein-protein interactions. These interactions do not resemble classical receptor-ligand interactions, but are rather complex protein-protein interactions with induced conformational changes. In addition, VWF comprises many different domains for protein-protein interactions and even domain A1, which is crucial for binding to schistosome eggs, binds to a large variety of structures, such as glycoprotein Ib (GPIb), Staphylococcus protein A, Snake Venom Metalloproteinase, collagen and heparin (156). For these reasons, the molecular mechanisms by which VWF binds to most other well-known interacting partners has not yet been resolved, and this prohibits the identification of potential VWF binding sites in the schistosome eggshell on the basis of structural homology to a known target.
In conclusion, our data demonstrated that eggshells of S. mansoni bind a specific set of plasma proteins, including VWF. We suggest that binding of plasma proteins to eggshells is physiologically relevant and a crucial step in extravasation as it promotes binding to damaged or activated endothelium. It thereby prevents eggs being swept into the circulation and it facilitates egg extravasation from the blood vessel, which is the first step in excretion of the egg.
Acknowledgements
We thank Irina Dragan (Center for Proteomics and Metabolomics, Leiden University Medical Center, Leiden, The Netherlands) for expert technical support.
Supplementary data
| Band number |
Plasma | Eggs1 | Eggs2 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Protein name | PC | TP | UP | Protein name | PC | TP | UP | Protein name | PC | TP | UP | |
| 1 | Albumin | 21.8 | 19 | 14 | Fibronectin 1 | 20.5 | 47 | 29 | Fibronectin 1 | 32.0 | 112 | 56 |
| 1 | Inter-alpha (Globulin) inhibitor H2 | 4.3 | 5 | 4 | Fibrinogen alpha chain | 6.9 | 6 | 6 | ||||
| 1 | Collagen, type II, alpha 1 | 4.8 | 6 | 5 | ||||||||
| 2 | Serum albumin | 25.3 | 22 | 14 | Fibronectin 1 | 38.4 | 160 | 66 | Fibronectin 1 | 30.9 | 117 | 51 |
| 2 | Alpha-2-macroglobulin | 8.0 | 11 | 11 | Talin 1 | 5.5 | 9 | 8 | Talin 1 | 3.4 | 5 5 | |
| 2 | Fibronectin 1 | 7.1 | 13 | 11 | ||||||||
| 3 | Serum albumin | 36.5 | 36 | 21 | Fibronectin 1 | 11.8 | 26 | 20 | Fibronectin 1 | 15.2 | 42 | 30 |
| 3 | Alpha-2-macroglobulin | 15.8 | 25 | 22 | Collagen, type II, alpha 1 | 2.9 | 6 | 4 | Collagen, type II, alpha 1 | 2.9 | 4 | 4 |
| 3 | Ceruloplasmin (ferroxidase) | 11.5 | 11 | 10 | Talin 1 | 1.5 | 4 | 4 | ||||
| 3 | Pregnancy zone protein | 8.4 | 6 | 6 | ||||||||
| 3 | Complement C3 | 3.7 | 5 | 4 | ||||||||
| 4 | Fibronectin | 1 2.8 | 7 | 5 | Fibronectin 1 | 4.9 | 11 | 8 | ||||
| 4 | Fibrinogen alpha chain | 4.7 | 4 | 4 | ||||||||
| 5 | Serum albumin | 49.6 | 64 | 29 | ||||||||
| 5 | Serotransferrin | 33.4 | 26 | 21 | ||||||||
| 5 | Ig mu chain C region | 18.1 | 8 | 7 | ||||||||
| 5 | Complement C3 | 4.9 | 5 | 5 | ||||||||
| 6 | Serum albumin | 75.9 | 132 | 46 | Fibrinogen alpha chain | 30.6 | 49 | 28 | Serum albumin | 32.8 | 21 | 16 |
| 6 | Fibrinogen alpha chain | 22.5 | 15 | 14 | Albumin (Fragment) | 22.7 | 6 | 5 | Fibrinogen alpha chain | 30.9 | 65 | 31 |
| 6 | Uncharacterized protein | 12.8 | 8 | 7 | Vitronectin | 19.7 | 14 | 9 | Vitronectin | 16.7 | 14 | 8 |
| 6 | Fibronectin 1 | 3.9 | 5 | 5 | Fibronectin 1 | 4.9 | 9 | 8 | ||||
| 6 | Collagen, type II, alpha 1 | 3.5 | 5 | 4 | ||||||||
| 7 | Alpha-1-antitrypsin | 51.2 | 28 | 17 | Fibrinogen beta chain | 41.1 | 33 | 22 | Fibrinogen beta chain | 35.6 | 27 | 19 |
| 7 | Serum albumin | 40.1 | 42 | 21 | Fibrinogen gamma chain | 30.2 | 17 | 12 | Uncharacterized protein | 19.5 | 8 | 7 |
| 7 | Ig gamma-1 chain C region | 36.1 | 14 | 10 | Fibrinogen alpha chain | 27.0 | 27 | 22 | Fibrinogen alpha chain | 19.2 | 19 | 16 |
| 7 | Fibrinogen beta chain | 22.8 | 14 | 10 | Albumin | 20.7 | 14 | 9 | Fibrinogen gamma chain | 16.5 | 8 | 6 |
| 7 | Ig alpha-1 chain C region | 15.6 | 6 | 4 | Vitronectin | 16.7 | 7 | 6 | Fibronectin 1 | 5.1 | 6 | 6 |
| 7 | Fibronectin 1 | 5.8 | 9 | 9 | ||||||||
| 8 | Fibrinogen gamma chain | 42.7 | 34 | 17 | Fibrinogen gamma chain | 34.7 | 30 | 13 | ||||
| 8 | Fibrinogen alpha chain | 23.2 | 20 | 16 | Actin, beta | 19.1 | 7 | 7 | ||||
| 8 | Actin, beta | 19.1 | 6 | 6 | Fibrinogen alpha chain | 16.7 | 16 | 12 | ||||
| 8 | Fibrinogen beta chain | 13.2 | 4 | 4 | Fibrinogen beta chain | 16.5 | 6 | 6 | ||||
| 8 | Albumin | 12.5 | 9 | 7 | Uncharacterized protein | 10.0 | 7 | 6 | ||||
| 8 | Fibronectin 1 | 9.7 | 18 | 16 | ||||||||
| 9 | Serum albumin | 25.1 | 19 | 13 | Actin, beta | 37.0 | 31 | 19 | Fibronectin 1 | 7.0 | 12 | 9 |
| 9 | Apolipoprotein A-IV | 21.7 | 10 | 8 | Actin, beta | 32.2 | 22 | 17 | ||||
| 9 | Fibrinogen alpha chain | 21.7 | 14 | 12 | Apolipoprotein A-IV | 22.7 | 13 | 9 | ||||
| 9 | Fibronectin 1 | 6.5 | 12 | 10 | Fibrinogen alpha chain | 9.8 | 5 | 5 | ||||
| 9 | Fibronectin 1 | 2.9 | 7 | 6 | ||||||||
| 10 | Serum albumin | 26.6 | 25 | 14 | Fibrinogen alpha chain | 4.2 | 8 | 4 | Fibrinogen gamma chain | 10.2 | 11 | 6 |
| 10 | Haptoglobin | 19.4 | 16 | 8 | Clusterin | 9.8 | 13 | 7 | ||||
| 10 | Apolipoprotein A-IV | 10.1 | 4 | 4 | Fibrinogen alpha chain | 8.5 | 11 | 6 | ||||
| 10 | Fibrinogen alpha chain | 5.5 | 7 | 5 | Fibronectin 1 | 2.8 | 6 | 6 | ||||
| 10 | Complement C3 | 2.3 | 4 | 4 | ||||||||
| 11 | Albumin | 10.2 | 8 | 5 | Apolipoprotein E | 24.3 | 10 | 7 | Apolipoprotein E | 26.8 | 9 | 8 |
| 11 | Clusterin | 15.4 | 17 | 8 | Clusterin | 16.7 | 12 | 9 | ||||
| 11 | Fibrinogen alpha chain | 9.7 | 10 | 7 | Fibrinogen alpha chain | 11.0 | 8 | 6 | ||||
| 11 | Fibronectin 1 | 3.0 | 7 | 6 | Fibrinogen gamma chain | 10.2 | 8 | 5 | ||||
| 12 | Apolipoprotein E | 19.9 | 6 | 6 | Apolipoprotein E | 42.3 | 36 | 15 | Apolipoprotein E | 34.7 | 31 | 13 |
| 12 | Serum albumin | 18.7 | 9 | 8 | Fibrinogen alpha chain | 11.9 | 7 | 5 | Fibrinogen alpha chain | 10.7 | 12 | 8 |
| 12 | Fibrinogen alpha chain | 6.1 | 6 | 4 | Fibrinogen gamma chain | 10.2 | 9 | 6 | Fibrinogen gamma chain | 10.6 | 10 | 6 |
| 12 | Fibronectin 1 | 3.4 | 12 | 8 | ||||||||
| 13 | Uncharacterized protein | 17.2 | 12 | 8 | Fibronectin 1 | 3.0 | 5 | 5 | ||||
| 14 | Albumin | 19.9 | 11 | 10 | Actin, beta | 12.3 | 4 | 4 | Tyrosine 3-monooxygenase/ tryptophan 5-monooxygenase activation protein, zeta polypeptide (Fragment) | 22.8 | 5 | 4 |
| 15 | Apolipoprotein A-I | 24.1 | 11 | 4 | ||||||||