Chapter 3
Schistosoma mansoni: The egg, biosynthesis of the shell and interaction with the host
Affiliations
- C1
- Department of Medical Microbiology and Infectious Diseases, Erasmus MC, Rotterdam, The Netherlands
Abstract:
The schistosome eggshell is a hardened and tanned structure made from cross-linked proteins. It is synthesized within the female worm from many different kinds of proteins and glycoproteins. Once the egg is released in the circulation, the outer surface of the eggshell is exposed and hence a direct site of interaction between the parasite and the host. The major eggshell protein is p14, but about one third of the eggshell is made from common cellular proteins, some of which are known to be immunogenic. This has many consequences for parasite-host interactions. However, so far, the eggshell has gained little attention from researchers. We will discuss the structure of the eggshell and its role in granuloma formation, host factor binding and egg excretion.
Introduction
Production of the Schistosoma egg
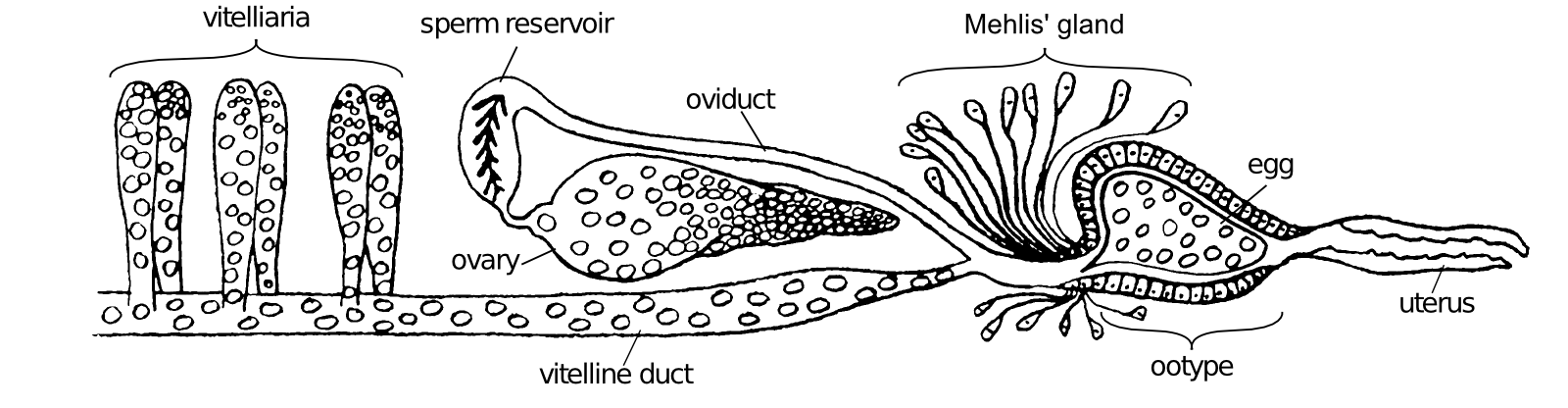
Schistosomes are digenetic parasitic flatworms. The female Schistosoma mansoni excretes around 350 eggs daily which equals to approximately one egg every 5 min (32). The oocyte is produced in the ovary and released in the oviduct (Fig. 3.1). Here it is fertilized by sperm that comes from the sperm reservoir, a dilated region in the oviduct. The fertilized oocyte moves further along the oviduct, which joins the vitelline duct. Here, 30-40 vitelline cells originating from the vitelline gland surround the fertilized oocyte (175). Together, the fertilized oocyte and its surrounding vitelline cells move to the ootype (Fig. 3.2a), thereby passing Mehlis' gland. The actual function of this structure remains elusive. Several functions of the Mehlis' gland have been suggested. It is proposed to lubricate the uterus for the passage of the egg or to activate spermatozoa. Other suggested functions of Mehlis' gland are involved in eggshell formation: release of eggshell granules, control or initiate cross-linking of the eggshell or provide a membrane which serves as a template on which the proteins accumulate to form the eggshell (174). Once in the ootype, contractions of the ootype make the vitelline cells release their granules which contain eggshell precursor proteins (Fig. 3.2b). The eggshell formation starts here. The eggshell is shaped by the ootype and strengthened through tyrosinase activity that causes cross-linking of the released eggshell precursor proteins (Fig. 3.2c and d). The egg is now ready to pass the uterus and to be released in the circulation, where the miracidium further matures within the eggshell (Fig. 3.2e).
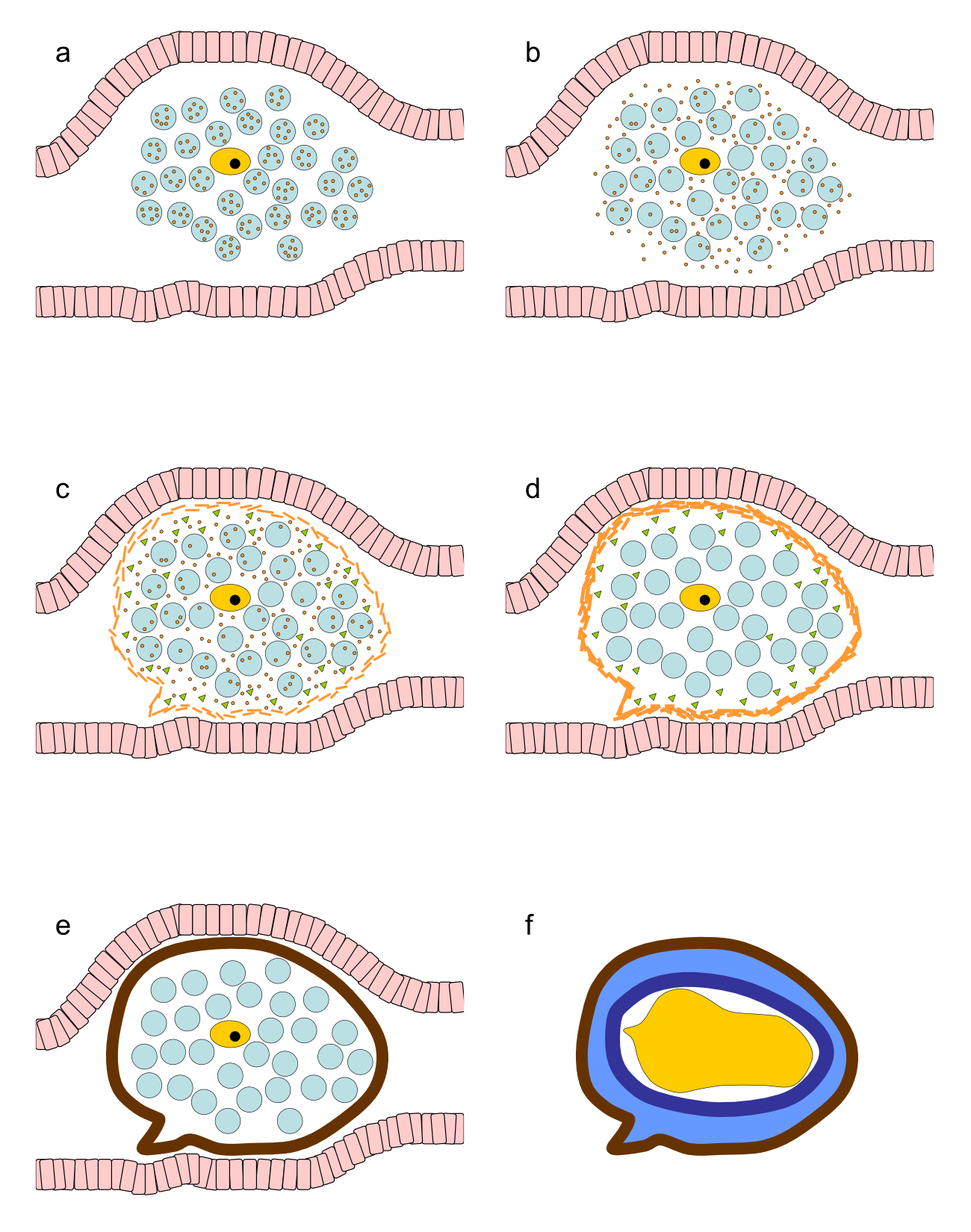
Maturation of this newly produced egg of S. mansoni, that simply consists of the cross-linked eggshell surrounding the ovum and vitelline cells, takes about a week. In this period the vitelline cells provide nutrients for the developing miracidium, which also obtains nutrients from the host (6). Newly deposited eggs lack complex subshell structures between the eggshell and the embryo, but these appear during the development of the egg (6,123). First, in an early stage, a few cells detach from the embryo and form a thin syncytial layer, known as "von Lichtenberg's envelope", between the eggshell and the developing miracidium (6,123). As development proceeds, this envelope becomes thicker, completely encloses the egg contents, the embryo and vitelline cells and separating them from the shell of the egg. This nucleated envelope contains extensive rough endoplasmic reticulum indicating that this is a place of active protein synthesis (6). During the further development a new layer of extra cellular material is formed between von Lichtenberg's envelope and the eggshell, called Reynolds' layer, which mainly consists of granulated material most likely originating from the envelope (6,123). The envelope and not the miracidium produces the proteins secreted by the egg, including IPSE/alpha-1 (109,162).
Eggs are produced one by one by female S. mansoni and the next egg will be produced once the previous one has been released. With an egg production of 350 per day, female schistosomes can be considered as true egg factories. This is reflected in the fact that about 10% of total worm mRNA encodes for one eggshell protein, p14 (128,170).
Structure of the eggshell
Cross-linking and tyrosinase activity
The eggshell is a hardened and tanned structure made from cross-linked proteins. The cross-linking process is known as quinone tanning and occurs by formation of quinone bonds. It is dependent on tyrosinase activity. Tyrosinases are copper-containing glycoenzymes that can catalyze both the hydroxylation of mono-phenols (tyrosine residues) to ortho-diphenols (L-dihydroxyphenylalanine, L-DOPA) and the subsequent oxidation of this L-DOPA to ortho-quinone (Fig. 3.3). In this way, accessible tyrosine residues of proteins are converted to o-quinones. These o-quinones are very reactive compounds and can form adducts by reaction with nucleophilic compounds, such as free amino or sulfhydryl groups on adjacent proteins. These reactions are known to occur in processes as diverse as the browning of foods and plant leaves, and the sclerotization and tanning of insect cuticles (11). Nowadays it is known that in plants and insects the formation of these quinone adducts occurs via two different reactions of free amino groups with the o-quinones: the formation of N-quinonyl derivatives (Michael-type adducts) and of Schiff bases (Fig. 3.3). Although it was originally suggested that in S. mansoni the cross-linking of the eggshell occurs via formation of the N-quinolyl adducts only (174), it is in our opinion more likely that also in helminths, the eggshell tanning occurs via both processes (Fig. 3.3).
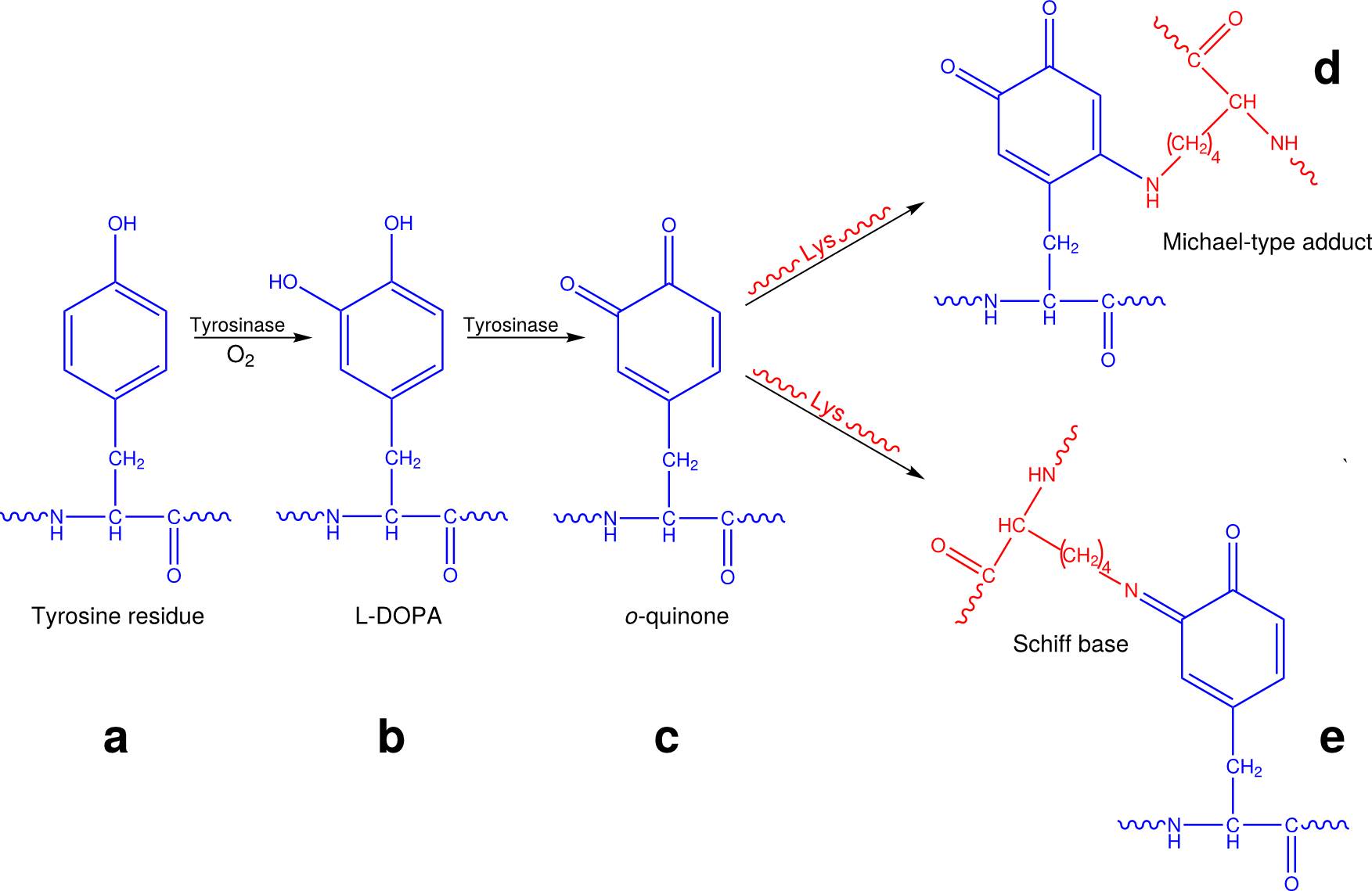
This cross-linking within a single protein and between neighboring proteins, results in an extensively cross-linked protein matrix. The appearance of the o-quinones leads to a rapid change in color and hardening of the proteins, rendering them intractable to all protease activity and only hydrolytic treatments can dissolve the cross-linked eggshell (194). The cross-linked eggshells are highly fluorescent. Their excitation/emission spectra are similar to the spectra of products formed by mushroom tyrosinase in a mixture of lysine with L-DOPA, indicating that in the eggshell lysine is the most probable amino acid that forms quinone bonds with tyrosine (see below) (165).
Tyrosinase activity appears to be developmentally regulated with peak transcription in mature female worms. Enzymatic activity is specifically located in the vitelline cells (56,65). Eggshell formation by tyrosinase activity is pH dependent and triggered by alkaline environment (195).
Specific eggshell proteins
Putative eggshell proteins have been identified in the late 80s by searching for specific mRNAs that are only expressed in mature females and not in males, nor in immature female worms, miracidia or cercariae. The corresponding genes could either encode eggshell precursors or proteins involved in the synthesis of the eggs and eggshell. Interestingly, tyrosinases, which are known to be developmentally regulated and involved in eggshell synthesis, have not been identified this way. This suggests that the catalytic enzymes required for eggshell formation are not abundantly expressed when compared to structural proteins. In addition, eggshell proteins that are not exclusively produced in mature females can not be detected this way.
The first putative eggshell protein to be described was p14 (13,14,93,94,150,169). There are several copies of the p14 gene in the genome of S. mansoni. They do not rearrange, and they are not sex linked (14,169). The p14 gene is developmentally regulated and only expressed in mature female worms. The p14 gene is only expressed in mature vitelline cells in the vitelline duct and within the egg enclosed by the eggshell in the ootype. Transcripts of p14 are undetectable in RNA obtained from eggs from the liver (33). The p14 gene is the most abundant mRNA transcript in female worms and accounts for 10% of the mRNA of the entire organism. Even in samples of mixed sex, this mRNA is by far the most abundant transcript (128). The p14 protein is present in all the vitelline droplets of vitelline cells in mature female worms (93). Immunoblots confirmed the presence of p14 in eggshell (47).
Although less abundant, p48, another putative eggshell protein, shows a similar expression pattern when compared to p14. However, the amount of p48 mRNA in worm is 10-30-fold less abundant than that of p14 (33). In vitro translation of the cDNA with [3H]tyrosine produces a 48 kDa protein. A protein of the same size is also present in translation products of mRNA from mature female worms, but not in that of male or immature female worm (88). Antibodies raised against recombinant p48 proteins recognize a 48 kDa protein on Western blot in homogenates of mature female worms, whereas no signal can be detected in homogenates of male worms or immature female worms. In addition, antisera raised against mature female worm extract recognize recombinant p48 proteins, while antisera raised against male or immature female worm extracts do not (33).
Reis et al. (1989)(144) described another female specific mRNA (FS800), that is only expressed in vitelline cells of mature female worms. The mRNA sequence was described to comprise two Open Reading Frames (ORFs) in two distinct reading frames. The longest ORF with the starting codon upstream of the alternative ORF is currently annotated as the translated protein sequence in the S. mansoni genome (9). However, to our knowledge, there are no data available yet on the translation of this female-specific mRNA into protein(s).
Finally, one other female specific protein has been described by Menrath et al. (1995)(117) and was described as mucin-like. Later, it was renamed p19 (119) but it has now been annotated as eggshell protein (Q26570_SCHMA) in the UniProt database. However, expression of the gene can be found in the epithelium surrounding the female oviduct close to its entrance into the ootype, but not along the vitelline duct and neither in ovary, vitellarium or ootype (117). Although this protein is female specific, this is apparently not an eggshell protein.
Non-specific eggshell proteins in the eggshell
Recently, the protein composition of purified eggshell fragments was investigated by a proteomic approach (47). This study showed that the eggshell is not only composed of the above mentioned specific eggshell proteins. About one third of the proteins in the eggshell are common cellular proteins. Among them are known schistosome antigens such as major egg antigen p40 and HSP 70, but also structural cellular proteins (actin and tubulin) and non-structural proteins of which many are glycolytic enzymes (47). Many of these non-specific proteins in the eggshell have also been identified inside the egg (110), in adult worm tegument (17,184) and in schistosomula and cercariae (40).
The list of non-specific eggshell proteins may not be complete, as proteins that are cross-linked are not always detectable by mass spectrometry (47). Although the incorporation of these non-specific eggshell proteins may be an unintended feature of eggshell production, the presence of these proteins, even in small amounts, may have immunological consequences. Many of them elicit a cellular or humoral immune response in the host upon a schistosome infection. Some of the incorporated proteins, such as glutathione-S-transferase (GST) and glyceraldehyde-3-phosphate dehydrogenase (GAPDH), were proposed and tested as vaccine candidates, albeit with little success in preventing infection (133). Vaccination with such antigens might actually induce pathology as it enhances the immune response against antigens in the eggshell, thereby enhancing granuloma formation and disease symptoms.
It should be noted that IPSE/alpha-1 and Omega-1, the two major egg secretion proteins (ESP), have not been found among the proteins of the eggshell (47). This indicates that the proteins excreted by eggs are not incorporated into the shell of the egg, which suggests that cross-linking of the eggshell proteins is finished before excretion of proteins by the egg begins.
Amino acid composition of the eggshell and eggshell proteins
In p14, 44% of the amino acids are glycine. This protein has some homology with the carboxyl terminal of mouse cytoskeleton keratin and with silk moth chorion proteins (13). The other major eggshell protein, p48, has only slightly elevated glycine levels, but contains high levels of lysine and aspartate, and its most abundant amino acid is tyrosine. The other proteins we identified as part of the eggshell do not have extreme levels of glycine, although their average glycine content is significant higher than the average glycine content of S. mansoni proteins (47).
The amino acid composition of fluorescent granules in the vitelline cells is highly similar to that of hydrolyzed eggshells, suggesting that all the reagents for eggshell formation are present in the vitelline cells (195). Hydrolyzed total eggshells revealed that 36% of the amino acids are glycine, which hereby largely attributes to the eggshell (24,47,150). The high glycine levels in the analysed eggshell hydrolysates suggest that p14 widely contributes to the eggshell. This contribution was estimated to be 70%. Immunoblots with anti-p14 antibodies showed that p14 is indeed present in eggshell (47). The role of the high glycine content of p14 and total eggshell remains elusive, but a high glycine content is associated with coiled coils. Rodrigues et al. (1989)(150) proposed a structure model of p14 based on its amino acid sequence and the high glycine content. In this structure, the protein is composed of short anti-parallel beta strands in which glycines and other residues with small side chains lie within the strands and tyrosines are arranged at the bends. This way, tyrosine residues are still available for cross-linking.
As tyrosine is essential in quinone tanning, eggshells are expected to contain high levels of this amino acid too. This is indeed the case for the two main eggshell proteins p14 (11.3%) and p48 (26.1%). However, little tyrosine can be measured in hydrolyzed eggshells or granules. This is probably not because of the absence of tyrosine, but due to the changes of the tyrosine molecule upon cross-linkage during eggshell formation, which makes them no longer recognizable as tyrosine (Fig. 3.3). Although little tyrosine could be detected in eggshell hydrolysates or fluorescent granules, radiolabeled tyrosine was efficiently incorporated in eggshell when injected in infected mice, indicating that eggshell does contain significant amounts of tyrosine (24).
A third amino acid that is expected to be abundant in eggshell is lysine, as this amino acid is likely to be the residue that forms the cross-link with the o-quinones (165). Indeed, the eggshell proteins p14 and p48 contains high levels of lysine (5.6% and 17.1%). However, no abundant levels of lysine could be detected in total eggshell. As with tyrosine, this may be due to cross-linking of the lysine residues to the o-quinones.
Glycans
The eggshell does not only consist of cross-linked proteins, but on these proteins, schistosome specific glycans are present. Composition analysis of glycans was suggestive for mannose and fucose residues and the presence of N-linked glycans, but additional O-linked glycans and other glycan classes may also be present. The glycan epitopes F-LDN(-F) and Lewis X are present on eggshell, as demonstrated by immunoblots (47). These glycans are known to be immunogenic, but little is known about the molecular basis of glycan-mediated host-parasite interactions (186).
Interaction with the host
Egg secretion proteins (ESP)
Although the eggshell is a direct site of interaction between the parasite and the host, little research has focused on this tightly cross-linked and hard to analyze structure. Rather, much research has focused on ESP. Secretion of ESP by the miracidia starts a few days after the eggs have left the female worm (6). There have been some contradictory papers reporting that ESP contains either a wide range of proteins (30) or only a few (6,110). There is, however, consensus that the major ESP are IPSE/alpha-1 and Omega-1. The total ESP and isolated or recombinant IPSE/alpha-1 or Omega-1 have widely been investigated for their capacity to drive a strong Th2 response. This immune response protects the host from fatal disease. However, this Th2 response is a strong inducer of granuloma formation (134). Furthermore, T-cell responses are essential for excretion of eggs. Experiments in mice have shown that in the absence of T-cells, egg excretion from the body is greatly hampered (49). In humans, HIV patients with low CD4+ T-cell count have reduced numbers of egg excretion (90).
Granuloma formation and the eggshell
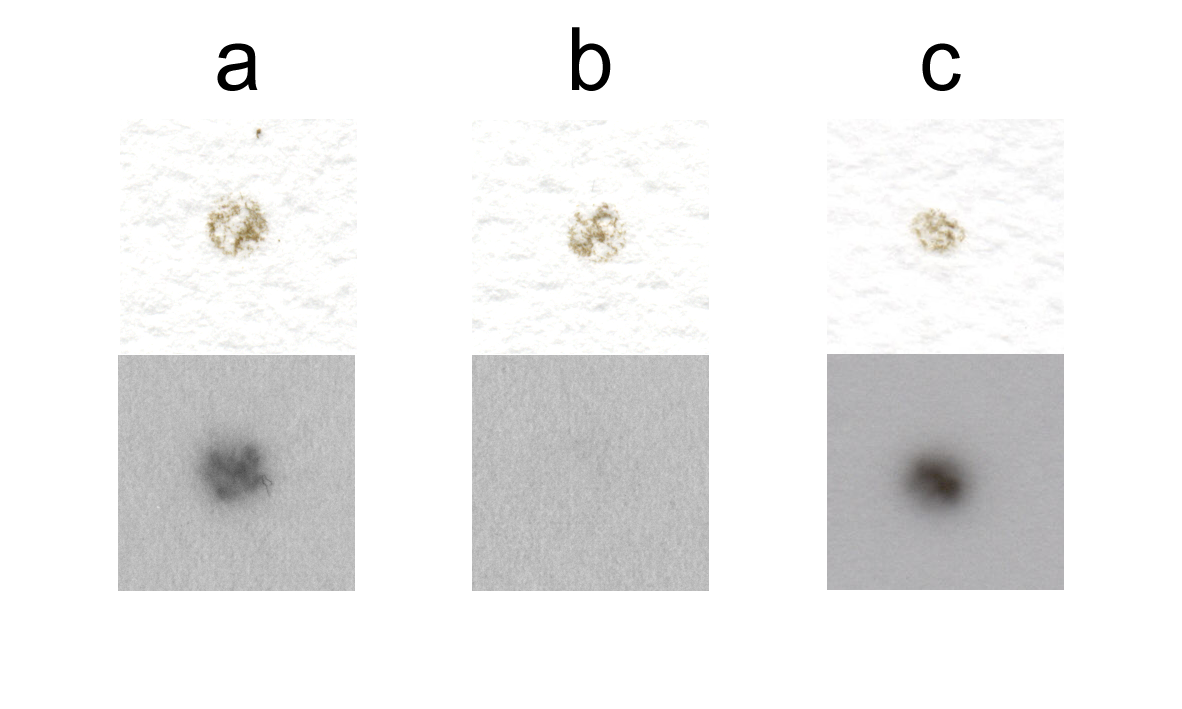
While many investigators have focused on ESP, much less research has been done regarding the eggshell. It has recently been shown that the eggshell contains numerous other proteins and glycoproteins besides the major eggshell proteins. Many of these other proteins and glycoproteins are known schistosome antigens, such as p40, phosphoenolpyruvate carboxykinase (PEPCK) and thioredoxin peroxidase. These antigenic proteins induce cellular responses (4, 200) or antibody responses (122). Serum of patients infected with schistosomes contains antibodies that recognize purified eggshells, from which all not covalently bound compounds were removed (47) (Fig. 3.4a), whereas these were not present in control serum (Fig. 3.4b). It is thus evident that not only ESP interact with the immune system, but also the eggshell itself is recognized by antibodies. Furthermore, eggs isolated from livers of infected hamsters are covered by host antibodies (Fig. 3.4c).
It is known that Th2-cells play a role in granuloma formation. Likewise, antibodies bound to the eggshell may enhance pathology. Since the eggshell is resistant to protease activity, and because the incorporated antigenic proteins in the eggshell bind host antibodies, the eggshell surface will form an indigestible antigen-antibody complex. These structures are known to induce chronic inflammatory responses that can lead to granuloma formation by a complex interaction between CD4+ T-cells, macrophages and cytokines (201). Also ESP are involved in granuloma formation around the egg (110,134).
Activation of T-cells and binding to antibodies may also play a role in the process of extravasation through the endothelium and the gut wall. Ex vivo, antibodies bind to ESP that exit the egg through pores in the eggshell. This is known as the circumoval precipitin (COP) reaction. At the point of secretion, a precipitate will form. Globular blebs or septate precipitates appear on the wall of the schistosome eggs after incubation with the serum of infected individuals (66).
How to get out of the host?
The egg is not capable of moving by itself. Thus, all movement has to be induced by external forces. First it is released into the circulation by the female worm; next the circulating blood will take hold of the egg. However, the intended pathway for the S. mansoni egg is not to follow the blood stream, but to extravasate the vessel wall and penetrate the gut wall in order to get out with the feces. This process takes at least six days, but may also take weeks (89). It is unlikely that the female worm can push such an enormous egg through the host structures. Therefore, the egg probably attracts host proteins and host cells in order to hold onto the vessel wall and help passage through the endothelium and the gut wall, allowing escaping the host and continuing the life cycle.
In addition to the widely studied schistosome-specific glycoproteins IPSE/alpha-1 and Omega-1, some house-keeping enzymes have been described to be virulence factors. Enolase and GAPDH, proteins that have also been identified as part of the eggshell, are such house-keeping enzymes which are also virulence factors in Schistosoma and other organisms. They both act as surface associated receptors that bind plasminogen, thereby capturing fibrinolytic activity of the host on the eggshell surface (206). This activity represents a mechanism to enhance virulence possibly required for tissue invasion and may thereby facilitate the escape of the eggs (132). Enolase in other pathogens has also been shown to bind actin and fibronectin. Fibronectin is a main component of extracellular matrix surrounding epithelial and endothelial cells. Blocking enolase decreased adhesion of other pathogens to endothelial cells (10,57). By binding to and activation of host factors, the eggshell may thus play an important role in getting out of the host. Thrombocytopenia significantly reduces egg excretion (125). Platelets massively adhere to eggshells of S. mansoni, resulting in activation of platelets (203). Activated platelets generally activate endothelial cells and may thereby facilitate passage through the endothelium. The eggshell may thus play an important role in getting out of the host by binding to and activation of host factors like plasminogen and platelets. Additionally, escape from the host may be facilitated by proteolytic activity of eggs and miracidia or from other (host) proteins and cells attracted by the egg (138,172).
Acknowledgments
Production of Fig. 3.3 by Michiel Bexkens is gratefully acknowledged. We sincerely thank the two anonymous reviewers for their helpful comments.