- Introduction
- Materials and methods
- Isolation of eggs
- Purification of eggshells
- Trypsin digestion
- HPLC and MS
- Database searching
- Criteria for protein identification
- Scanning electron micrography with immunogold nanoparticle labeling
- Western blots
- Monosaccharide composition analysis
- Amino acid analysis of eggshell fragments
- Results
- Purification of the eggshells
- Analysis of the eggshell proteome
- Analysis of eggshell glycoconjugates
- Analysis of the eggshell amino acid composition
- Immune reactivity of the eggshell proteins
- Discussion
- Acknowledgements
Chapter 2
The proteome of the insoluble Schistosoma mansoni eggshell skeleton
Affiliations
- B1
- Department of Medical Microbiology and Infectious Diseases, Erasmus MC, Rotterdam, The Netherlands
- B2
- Department of Biomolecular Mass Spectrometry, Bijvoet Center for Biomolecular Research and Utrecht Institute for Pharmaceutical Sciences, Utrecht University, Utrecht, The Netherlands
- B3
- Institute for Veterinary Research, IVW/GSAH, Utrecht, The Netherlands
- B4
- Department of Haematology, University Medical Center Utrecht, Utrecht, The Netherlands
- B5
- Department of Parasitology, Leiden University Medical Center, Leiden, The Netherlands
- B6
- Netherlands Proteomics Centre, Utrecht, The Netherlands
Abstract:
In schistosomiasis, the majority of symptoms of the disease is caused by the eggs that are trapped in the liver. These eggs elicit an immune reaction that leads to the formation of granulomas. The eggshell, which is a rigid insoluble structure built from cross-linked proteins, is the site of direct interaction between the egg and the immune system. However, the exact protein composition of the insoluble eggshell was previously unknown. To identify the proteins of the eggshell of Schistosoma mansoni we performed LC-MS/MS analysis, immunostaining and amino acid analysis on eggshell fragments. For this, eggshell protein skeleton was prepared by thoroughly cleaning eggshells in a four-step stripping procedure of increasing strength including urea and SDS to remove all material that is not covalently linked to the eggshell itself, but is part of the inside of the egg, such as Reynolds' layer, von Lichtenberg's envelope and the miracidium. We identified 45 proteins of which the majority are non-structural proteins and non-specific for eggs, but are house-keeping proteins that are present in large quantities in worms and miracidia. Some of these proteins are known to be immunogenic, such as HSP70, GST and enolase. In addition, a number of schistosome-specific proteins with unknown function and no homology to any known annotated protein were found to be incorporated in the eggshell. Schistosome-specific glycoconjugates were also shown to be present on the eggshell protein skeleton. This study also confirmed that the putative eggshell protein p14 contributes largely to the eggshell. Together, these results give new insights into eggshell composition as well as eggshell formation. Those proteins that are present at the site and time of eggshell formation are incorporated in the cross-linked eggshell and this cross-linking does no longer occur when the miracidium starts secreting proteins.
Introduction
Schistosomiasis is a tropical disease affecting an estimated 200 million people worldwide, mainly in sub-saharan Africa (75). The disease is caused by a parasitic flatworm of the Schistosoma genus which can reside in the veins of its host for many years. Apparently, the host fails to elicit an adequate immune response to the adult worms. In contrast, the eggs provoke a strong immune response and are the main cause of pathology in schistosomiasis. To complete the life cycle and be excreted with the faeces, the Schistosoma mansoni eggs have to extravasate from the vessel and penetrate through the intestinal wall, thereby damaging the tissues they pass. This process is dependent on an immune reaction which is elicited by the eggs (49). Furthermore, the eggshells appear to be potent inducers of platelet activation, which may facilitate extravasation (203). Although the eggs have to be excreted with the faeces to propagate the life cycle, many of the eggs do not reach the intestines. Instead, they follow the blood flow and are trapped in the liver, where they are responsible for granuloma formation which can lead to fibrosis and portal hypertension with all its complications in chronic disease. The granulomas are a result of massive inflammation around the eggs, due to the strong immune response that they induce. This immune response is skewed towards a T-helper 2 response, which is characteristic for helminth infections.
The Schistosoma eggshell is a porous protein structure with microspines on the outer surface (123). The proteins that form the eggshell are cross-linked by quinone tanning, making the eggshell a very rigid structure (194). This quinone tanning is a result of tyrosinase activity, which converts tyrosine residues into o-quinones. These o-quinones react with nucleophiles such as lysine and histidine, resulting in a series of cross-links within a single protein as well as between distinct proteins. Tyrosinase activity has been shown to be essential in eggshell formation (65).
Although the eggs play an important role in development of the disease, the protein composition of their shell, which is the site of direct interaction between the eggs and the immune system, has not yet been reported, even though female-specific proteins, as determined by mRNA expression or by labeled amino acid uptake, have been postulated to be putative eggshell proteins (14,33,88,144).
Recently, a study was published where several protein fractions of the Schistosoma egg were analysed by two-dimensional (2-D) gel electrophoresis and mass spectrometry (MS) (110). These five different fractions consisted of mature and immature eggs, miracidia, hatch fluid and egg secreted proteins (ESPs). In all of the fractions except ESP, the list of identified proteins was similar, containing a range of proteins from different functional categories. The ESP fraction appeared to contain just a small and unique subset of the total egg proteome, in strong contrast with a previous study where the list of proteins secreted by the eggs was similar to those from the other fractions (30). The only fraction of the S. mansoni egg of which the proteome has not been characterised is the insoluble shell of the egg, which is the actual site of direct interaction with the host.
A published proteomic analysis of eggshell of the closely related Schistosoma japonicum identified 520 proteins. The list of proteins of S. japonicum eggshell included a wide range of proteins such as putative eggshell proteins, previously characterised egg proteins, motor proteins, chaperones and enzymes (100). However, this list is probably too extensive as in these samples the eggshells were not purified from all attached material from the inside of the eggs. Proteins from the structures underlying the eggshell are not cross-linked and may be over-represented in the MS analysis as they are easier to detect and identify than the cross-linked eggshell proteins.
Despite its importance in pathogenicity and host-parasite interactions, the composition of the S. mansoni eggshell has not been determined. In this study we performed LC-MS/MS on purified eggshell fragments to identify the proteins of which the shell is composed. The results showed that the Schistosoma eggshell is not only made from known putative eggshell proteins, but from a range of proteins, some of which are known to be immunogenic. Identification of the eggshell proteins showed that apparently eggshell formation is completed before maturation of the egg.
Materials and methods
Isolation of eggs
A Puerto Rican strain of S. mansoni was maintained in Golden hamsters for which animal ethics was approved (licence EUR1860-11709). Animal care and maintenance was in accordance with institution and governmental guidelines. Eggs were isolated by overnight digestion of livers from infected hamsters in 500 ml 1.8% NaCl with 5 ml collagenase buffer (6.8 mM NaCl, 0.7 mM KCl, 9.2 mM HEPES, 0.8 mM CaCl2 and 745 U/ml collagenase Type A1 (Sigma, St Louis, MO, USA) pH 7.8) (166). Subsequently, for the isolation of eggs, the digested liver suspension was passed over sieves. Undigested liver material was removed by the first two sieves (425 and 180 μm mesh), after which the eggs were collected on the third (45 μm mesh). The eggs were then washed in Dutch spring water (Bar-le-Duc, Utrecht, The Netherlands) and allowed to hatch for at least 3 h. Eggshells were collected, frozen in liquid nitrogen and crushed in a micro-dismembrator S (Braun Biotech Int., Melsungen, Germany) by shaking at 2000 rpm for 2 min. Finally, the eggshell fragments were layered on a 60% (w/v) sucrose cushion and centrifuged at 650g for 2 min. Subsequently, the eggshell pellets were washed three times with MilliQ water to remove remaining sucrose.
Purification of eggshells
In order to remove all attached cellular material from the eggshells, the eggshell fragments were further purified in four sequential steps. First, the eggshell fragments were incubated in 2 M NaCl in PBS at room temperature for 30 min. Next, the eggshell fragments were incubated in 1% (v/v) Triton X-100 in PBS at room temperature for 30 min followed by incubation in 8 M urea with 0.6% β-mercaptoethanol in PBS at room temperature for 30 min. Finally, the eggshell fragments were incubated in 1% (w/v) SDS at 95 °C for 30 min. Eggshell fragments were washed three times after each of the first three purifications steps and five times after the final step.
After each purification step, eggshell fragments were collected for protein identification by LC-MS/MS after trypsin treatment.
Trypsin digestion
Laemmli buffer was added to the eggshell fragments and proteins were incubated at 37 °C for 30 min before loading on a 12% SDS-PAGE. The samples were run until the loading front reached the stacking/running gel interface. The gel was then fixed in 5% acetic acid 30% methanol and subsequently proteins were stained using GelCode Blue reagent (Pierce, Rockford, IL, USA). After destaining in MilliQ water, a single gel piece surrounding the loaded well (containing all proteins and eggshell fragments) was excised and subjected to in-gel trypsin digestion. For this, the gel piece was cut into small pieces, which were rinsed with MilliQ water and treated as described previously (184).
HPLC and MS
Peptides generated by in-gel digestion were analysed by nanoflow liquid chromatography using an Agilent 1100 HPLC system (Agilent Technologies, Waldbronn, Germany) coupled on-line to a 7-tesla LTQ-FT mass spectrometer (Thermo Electron, Bremen, Germany). The system was operated in a set-up essentially as previously described (116). ReproSil-Pur C18-AQ, 3 μm (Dr. Maisch, GmbH, Ammerbuch, Germany) was used as a resin for capillary reversed phase chromatography. Peptides were trapped at 5 μl/min on a 1 cm column (100-μm internal diameter, packed in-house) and eluted to a 15 cm column (50-μm internal diameter, packed in-house) at 150 nl/min on a 60 min gradient from 0% to 50% acetonitrile in 0.1 M acetic acid. The eluent was sprayed via emitter tips (made in-house) butt-connected to the analytical column. The mass spectrometer was operated in data-dependent mode, automatically switching between MS and MS/MS acquisition. Full scan MS spectra were acquired in Fourier-transform ion cyclotron resonance (FT-ICR) MS with a resolution of 20,000 at a target value of 2,000,000. The three most intense ions were then isolated for accurate mass measurements by a selected ion monitoring scan in FT-ICR with a resolution of 50,000 at a target accumulation value of 50,000. These ions were then fragmented in the linear ion trap using collision-induced dissociation at a target value of 15,000.
Database searching
Tandem MS were extracted, charge state deconvoluted and deisotoped by BioWorks version 2.0. All MS/MS samples were analysed using Mascot (Matrix Science, London, UK; version 2.2.1). Mascot was set up to search the GeneDB_Smansoni_Proteins.v4.0h, assuming the digestion enzyme trypsin and allowing for two missed cleavages. Mascot was searched with a fragment ion mass tolerance of 0.80 Da and a parent ion tolerance of 50 parts per million (PPM). Iodoacetamide derivative of cysteine was specified in Mascot as a fixed modification. Oxidation of histidine, methionine and tryptophan was specified in Mascot as a variable modification.
Criteria for protein identification
Scaffold (version 3.0, Proteome Software Inc., Portland, OR, USA) was used to validate MS/MS based peptide and protein identifications. Peptide identifications were accepted if they could be established at greater than 95.0% probability as specified by the Peptide Prophet algorithm (92). Protein identifications were filtered using a false discovery rate of <5% and were accepted if they could be established at greater than 99.0% probability and contained at least two identified peptides. Protein probabilities were assigned by the Protein Prophet algorithm (124). Proteins that contained similar peptides and could not be differentiated based on MS/MS analysis alone were grouped to one family to satisfy the principles of parsimony. Proteins that were identified in at least two out of three independent experiments were considered as true hits.
Scanning electron micrography with immunogold nanoparticle labeling
Egg or eggshell fragment samples were fixed in 2% glutaraldehyde (Merck, Germany)/HEPES prior to incubation with primary antibody against mouse heat shock protein 70 (HSP70); control samples were mock treated. Then, a second antibody goat anti-mouse 30 nm IgG-Gold nanoparticle (DGMGL-B001, BioAssay Works, LLC, Ijamsville, MD, USA) was used for recognition of the primary antibody. Samples were fixed for a second time with 3% glutaraldehyde/HEPES and subsequently dehydrated in a 80%, 90% and 100% graded series of ethanol and 1,1,1,3,3,3-Hexamethyldisilazane (MP Biomedicals Inc., Solon, Ohio, USA). The samples were sputter-coated with a thin layer of 6.5 nm platinum (spurt density:21.45) in a sputter coater and viewed in a scanning electron microscope (Philips XL30, Philips, Netherlands) with backscatter electron detector and secondary electron detector, Bias (volt) -216 V, Acc V 5.00 kV, work distance 2.9 and magnification 100,000×.
Western blots
Soluble egg antigens (SEA), adult worm antigens (AWA) and approximately 1 μg of schistosome GST and rabbit muscle enolase (Sigma) were run through a 12% SDS-PAGE. SEA and AWA were prepared by homogenisation in PBS followed by centrifugation. Purified protein standards (Fermentas, St. Leon-Rot, Germany) were loaded onto the gel. After separation, proteins were transferred to Polyvinylidene Fluoride (PVDF). Pooled sera from three infected hamsters were used in 1:100 dilutions as the primary antibody. Horseradish peroxidase (HRP) conjugated anti-hamster IgG (Abcam, Cambridge, UK) was used as a secondary antibody. Blots were developed using Enhanced Chemiluminescence (ECL) detection reagents (Pierce) on hyperfilm ECL (Amersham, GE healthcare, Diegem, Belgium) according to the manufacturers' instructions.
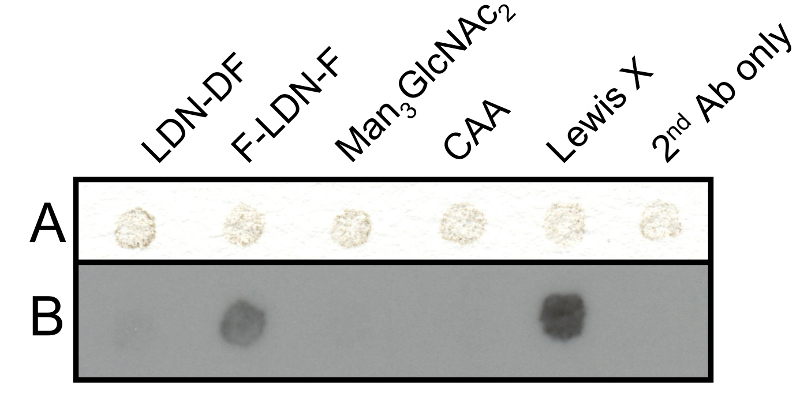
Glycan-specific monoclonal antibodies (mAbs) generated from schistosome-infected or immunized mice were obtained and characterised as previously described. The following mAbs were used to detect eggshell-associated glycans in dot blots: 114-5B1 (IgG1; binds to GalNAcβ1-4(Fucα1-2Fucα1-3)GlcNAc (LDN-DF) (192)); 128-1E7-C (IgM; binds to Fucα1-3GalNAcβ1-4(Fucα1-3)GlcNAc (F-LDN-F) and Fucα1-3GalNAcα1-4GlcNAc (F-LDN) (41,149)); 100-4G11 (IgM; binds to Man3GlcNAc2(41,191)); 120-1B10 (IgG1; binds to Circulating Anodic Antigen (CAA) (46)); 291-4D10 (IgM; binds to Galβ1-4(Fucα1-3)GlcNAc (Lewis X) (149,192)).
Rabbit antibodies against p14 were kindly provided by Prof. Dr. P.T. LoVerde (University of Texas Health Science Center, San Antonio, Texas, USA). Eggshell fragments were incubated with antibodies, washed three times and subsequently incubated with HRP conjugated anti-mouse or anti-rabbit immunoglobulin antibodies (Dako, Glostrup, Denmark). After washing, the eggshell fragments were incubated in ECL detection reagent and dotted on Whatman paper. Chemiluminescent signal was captured on hyperfilm ECL.
Monosaccharide composition analysis
A 35 μl aliquot of eggshell suspension was dried under a flow of nitrogen in a glass vial with insert. Dried eggshells were dissolved in 50 μl 4 M trifluoroacetic acid, hydrolysed by incubation at 100 °C for 4 h and subsequently dried under a flow of nitrogen. Fluorescent labeling of reducing monosaccharides generated by hydrolysis was performed following the method of Ruhaak et al. (2010)(157) with some slight modifications. Briefly, a solution of 23% acetic acid in DMSO was used to dissolve anthranilic acid (Sigma, Germany) to a concentration of 48 mg/ml and subsequently 2-picoline borane complex (Sigma, Germany) was added to a concentration of 107 mg/ml. The monosaccharides were dissolved in 10 μl of this labeling mix and reacted at 65 °C for 2 h. Labeled monosaccharides were diluted in 190 μl 0.6% sodium acetate.
Labeled monosaccharides were separated by reverse phase (RP)-HPLC on a Superspher 100 RP-18 endcapped column (250 × 4 mm; Grom, Germany). Solvent A consisted of 0.1% butylamine, 0.5% phosphoric acid and 1% tetrahydrofuran. Solvent B consisted of solvent A/acetonitrile 50/50.The following gradient conditions were applied: at time t = 0 min, 8% solvent B; at t = 5 min, 8% solvent B; at t = 30 min, 23% solvent B; at t = 32 min, 100% solvent B; at t = 42 min, 100% solvent B; at t = 43 min, 8% solvent B; at t = 60 min, 8% solvent B. The flow rate was 500 μl/min. Samples were injected in a 10× dilution with 0.6% sodium acetate. Fluorescence was detected at 360/420 nm. Monosaccharides were identified on the basis of their retention time compared with a reference monosaccharide mixture. The relative monosaccharide composition was calculated on the basis of fluorescence.
Amino acid analysis of eggshell fragments
The analysis of amino acid composition of purified eggshell fragments was performed by Ansynth service BV, Roosendaal, the Netherlands. Prior to analysis, the purified eggshell fragments were hydrolysed overnight in 6 M HCl at 110 °C. Analyses were performed using a Biochrom amino acid analyser equipped with a Ninhydrin detection system. The amounts of alanine, arginine, aspartate plus asparagine, glutamate plus glutamine, glycine, histidine, isoleucine, leucine, lysine, phenylalanine, proline, serine, threonine, tyrosine and valine were determined.
Results
Purification of the eggshells
The Schistosoma eggshell is a porous protein structure with microspines on the outer surface (123). The proteins that form the eggshell are cross-linked by quinone tanning, making the eggshell a very rigid structure (194). By light microscopy, the eggshells collected directly after digestion of livers appeared clean (Fig. 2.1A). However, those were expected to contain attached material from the underlying Reynolds' layer, von Lichtenberg's envelope and Lehman's lacuna (123). To ensure that all of this attached material would be removed, we purified the fragments of the eggshells in four consecutive steps with reagents of increasing strength. After crushing the eggshells, the eggshell fragments were treated consecutively with (i) 2 M NaCl to remove proteins attached exclusively by ionic strength, (ii) 1% (v/v) Triton X-100 to remove proteins attached by hydrophobic interactions, (iii) 8 M urea containing 0.6% β-mercaptoethanol and (iv) 1% SDS at 95 °C to remove all proteins that were linked non-covalently or by disulfide bonds. As the Schistosoma eggshell is a rigid structure of which the proteins are extensively cross-linked by quinone tanning, the eggshells were not dissolved after the final purification step by treatment with SDS and therefore eggshell fragments were still visible (Fig. 2.1B). We refer to these purified eggshell fragments as the protein skeleton of S. mansoni eggshells. Electron scanning microscopy showed that the structure of the eggshell appeared different after each purification step (Fig. 2.2). The eggshell surface directly after isolation is a three-dimensional (3-D) structure with spines and pores similar to previous descriptions (140,160). The surface became smooth after NaCl and Triton X-100 treatment of the eggshell fragments. After treatment with 8 M urea, the denatured proteins had swollen. This swelling was reduced after SDS treatment, where the eggshell skeleton was left as a wrinkled and more flattened structure.


Analysis of the eggshell proteome
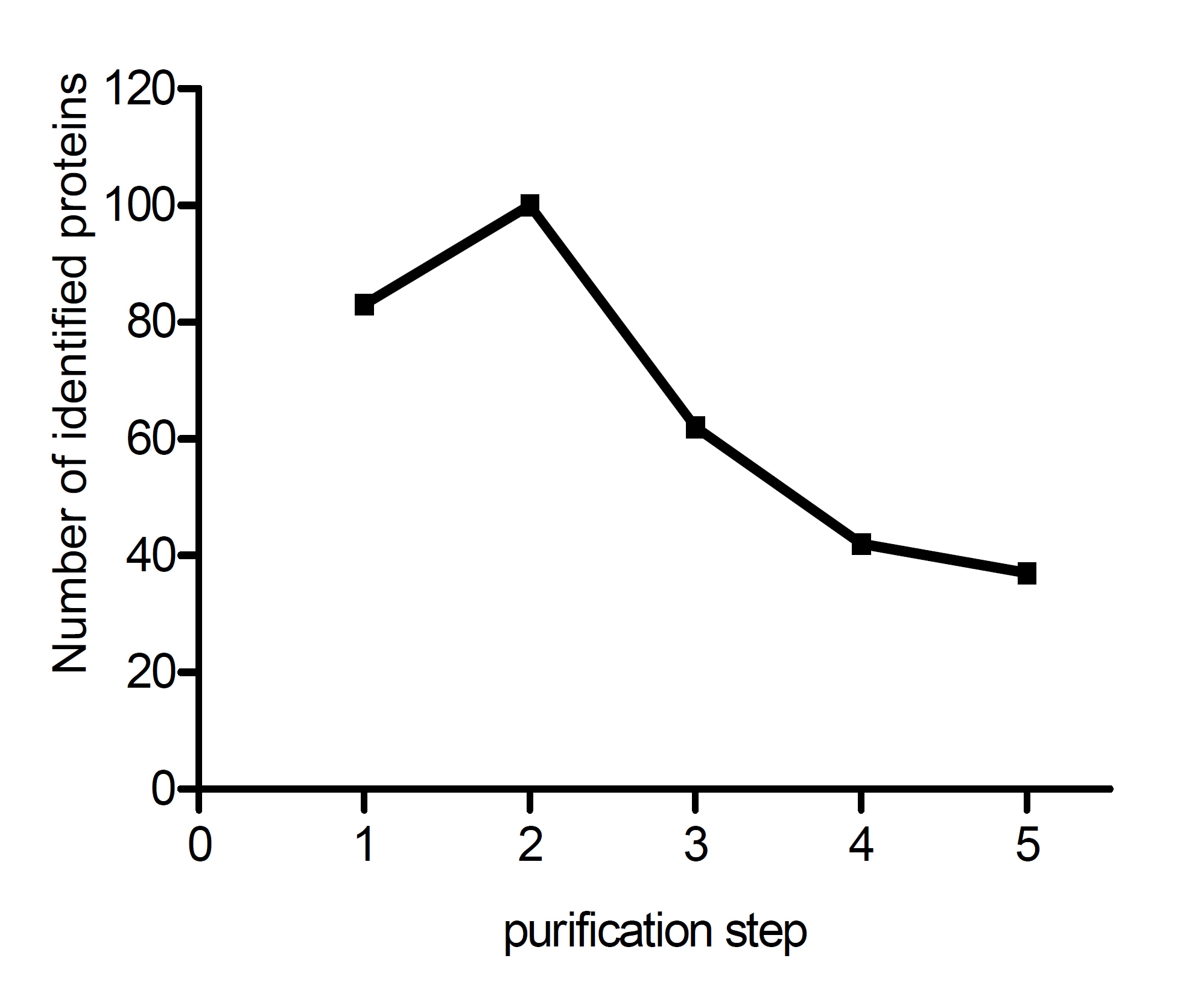
After each purification step, trypsin digests of the eggshell fragments were used for protein identification by LC-MS/MS. The number of identified proteins declined from 118 directly after eggshell isolation to a final total of 45 proteins in the protein skeleton (Fig. 2.3). The supernatant of the final wash step, after the SDS incubation, did not contain any proteins. The extensive washes after each purification step to remove the liberated proteins thus ensured that all of the identified proteins originated from the eggshell protein skeleton itself and are therefore part of the proteome of the cross-linked eggshell.
Among the identified eggshell proteins, we found some known schistosome antigens, such as major egg antigen p40 and HSP70 (38,121,176). In addition, multiple structural proteins that are normally part of the cytoskeleton of a cell were identified, such as actin and β-tubulin. Extracellular structural proteins, such as fibrin or collagen, were not identified. Furthermore, in this MS analysis we did not identify any of the putative eggshell proteins (see below in Sections Analysis of the eggshell amino acid composition and Discussion however).enough, the majority of the identified proteins were non-structural proteins. We identified membrane proteins, cytosolic proteins and nuclear proteins. A collection of enzymes was identified, of which many were glycolytic enzymes (Table 2.1). It is yet unknown whether the enzymes present in the eggshell protein skeleton are still catalytically active in situ. However, residual catalytic activity of enzymatic proteins in the eggshell is highly unlikely as these proteins are tightly cross-linked to other proteins and incorporated into the eggshell.
Protein identifications by MS were verified by localising one of the identified eggshell proteins on purified eggshell fragments by immunoscanning electron microscopy. Fig. 2.4 shows HSP70 on the eggshell fragments before and after purification. Its presence validates the protein identifications by MS.

Analysis of eggshell glycoconjugates
Schistosome eggs are known to contain and secrete glycoproteins that carry immunogenic glycans (85). We investigated whether glycan antigens were still present on the eggshell protein skeleton. Blotting of eggshells with a set of schistosome glycan reactive mAbs showed that mAb 128-1E7-C (weak) and 291-4D10 (strong) generate a positive signal, indicating that the glycan epitopes F-LDN(-F) and Lewis X are expressed on eggshell glycans, whereas the other epitopes tested are not expressed or not detectable (Fig. 2.5).
| Accession number | Protein name | Protein MW (kDa) | Number of unique peptides | Protein coverage (%) | |||
|---|---|---|---|---|---|---|---|
| Exp 1 | Exp 2 | Exp 3 | |||||
| Energy metabolism | |||||||
| Smp_002880.1 | ATP synthase alpha subunit mitochondrial | 60 | 5 | 13 | 2 | 30 | |
| Smp_038100 | ATP synthase beta subunit | 56 | 6 | 9 | 4 | 25 | |
| Smp_194770 | ATP:guanidino kinase (Smc74) | 95 | 2 | 13 | 0 | 21 | |
| Smp_024110 | Enolasea | 47 | 3 | 5 | 2 | 10 | |
| Smp_042160.2 | Fructose 1,6-bisphosphate aldolase | 40 | 0 | 4 | 2 | 15 | |
| Smp_056970.1 | Glyceraldehyde-3-phosphate dehydrogenase (phosphorylating) | 36 | 3 | 9 | 1 | 32 | |
| Smp_143840 | Glycogen phosphorylase | 80 | 2 | 8 | 0 | 14 | |
| Smp_038950 | Lactate dehydrogenase |
36 | 2 | 7 | 1 | 23 | |
| Smp_035270.2 | Malate dehydrogenase | 31 | 2 | 7 | 0 | 22 | |
| Smp_047370 | Malate dehydrogenase | 36 | 2 | 7 | 4 | 23 | |
| Smp_130300 | Na+/k+ atpase alpha subunit | 93 | 2 | 5 | 1 | 7 | |
| Smp_005880 | Phosphoenolpyruvate carboxykinase | 70 | 4 | 13 | 2 | 30 | |
| Smp_187370 | Phosphoglycerate kinase | 18 | 2 | 5 | 1 | 30 | |
| Smp_059790.2 | Transketolase | 63 | 2 | 4 | 0 | 9 | |
| Protein folding and stress response | |||||||
| Smp_073880.1 | 40S ribosomal protein S3A | 29 | 2 | 4 | 0 | 17 | |
| Smp_054160 | Glutathione S-transferase 28 kDa (GST 28) (GST class-mu) | 24 | 2 | 4 | 0 | 18 | |
| Smp_072330.2 | Heat shock protein | 81 | 6 | 25 | 9 | 38 | |
| Smp_106930.2 | Heat shock protein 70 | 69 | 17 | 30 | 5 | 46 | |
| Smp_069130.2 | Heat shock protein 70 (hsp70)-4 | 94 | 2 | 11 | 1 | 17 | |
| Smp_062420.1 | Heat shock protein 70 (hsp70)-interacting protein | 31 | 0 | 3 | 2 | 13 | |
| Smp_008545 | Heat shock protein HSP60 | 61 | 3 | 10 | 1 | 20 | |
| Smp_049250 | Major egg antigen p40 |
40 | 4 | 11 | 4 | 44 | |
| Smp_079770.1 | Protein disulfide-isomerase er-60 precursor (erp60) | 54 | 2 4 | 0 | 10 | ||
| Smp_095980 | Superoxide dismutase precursor (EC 1.15.1.1) | 20 | 9 | 3 | 3 | 44 | |
| Smp_059480 | Thioredoxin peroxidase | 21 | 3 | 4 | 1 | 25 | |
| Cytoskeleton | |||||||
| Smp_161920 | Actin | 42 | 5 | 16 | 5 | 51 | |
| Smp_090120.1 | Alpha tubulin | 50 | 7 | 13 | 2 | 30 | |
| Smp_085540.6 | Myosin heavy chain | 211 | 2 | 9 | 0 | 6 | |
| Smp_035760 | Tubulin beta chain | 50 | 13 | 23 | 7 | 51 | |
| Membrane proteins | |||||||
| Smp_020550 | Low-density lipoprotein receptor (ldl) | 87 | 7 | 9 | 4 | 12 | |
| Smp_159420 | Low-density lipoprotein receptor (ldl) | 103 | 4 | 4 | 0 | 5 | |
| Smp_179370 | Low-density lipoprotein receptor (ldl) | 94 | 3 | 3 | 1 | 6 | |
| Smp_091240.1 | Voltage-dependent anion-selective channel | 31 | 2 | 3 | 0 | 12 | |
| Protein synthesis | |||||||
| Smp_099870 | Elongation factor 1-alpha (ef-1-alpha) | 51 | 13 | 14 | 2 | 37 | |
| Smp_030690 | Elongation factor 1-beta | 24 | 2 | 2 | 0 | 11 | |
| Smp_143140 | Eukaryotic translation elongation factor | 22 | 1 | 2 | 2 | 15 | |
| Smp_143150 | Eukaryotic translation elongation factor | 61 | 2 | 7 | 0 | 17 | |
| Nuclear proteins | |||||||
| Smp_053290 | Histone H4 | 11 | 3 | 8 | 0 | 52 | |
| Other and unknown functions | |||||||
| Smp_179260 | Alpha-galactosidase/alpha-n-acetylgalactosaminidase | 109 | 5 | 10 | 0 | 13 | |
| Smp_174530 | Aminopeptidase PILS (M01 family) | 111 | 0 | 2 | 2 | 2 | |
| Smp_147890 | Rootletin (Ciliary rootlet coiled-coil protein) | 234 | 31 | 70 | 8 | 36 | |
| Smp_005860.1 | Expressed proteind | 47 | 4 | 7 | 4 | 21 | |
| Smp_148160 | Expressed proteine | 35 | 2 | 4 | 0 | 14 | |
| Smp_160560 | Expressed proteinf | 79 | 8 | 8 | 2 | 13 | |
| Smp_000270 | Hypothetical proteing | 26 | 2 | 0 | 3 | 21 | |
|
a Annotated as phosphopyruvate hydratase. b Annotated as malate dehydrogenase. c Annotated as heat shock protein. d Similar to heterogenous nuclear ribonucleoprotein k. e Similar to spermatogenesis-associated protein 6 precursor (SPATA6). f Similar to TyrA protein. g Similar to FS800, a female-specific protein. |
|||||||
To confirm the occurrence of glycans on eggshells, a monosaccharide composition analysis on hydrolysed eggshells was performed (Table 2.2). The composition analysis indicates the presence of a number of different constituent monosaccharides associated with the eggshells. The occurrence of mannose residues (three in every N-linked glycan core) suggests that N-linked glycans are present, but in addition O-linked glycans or other glycan classes would be in agreement with this overall composition. We have not been able to discriminate between xylose and fucose. In an N-linked glycan from schistosome eggs, up to one xylose residue in each N-glycan may be present. This suggests that at least fucose residues are also present, which is in line with the dot blot results showing staining for fucosylated glycan epitopes. At this stage it is not possible to derive any details about eggshell glycosylation, or which proteins are carrying the putative glycans.
| Monosaccharide | Relative ratios (Man set to 3) |
|---|---|
| Man | 3 |
| GlcNAc | 10 |
| Gal | 3 |
| GalNAc | 3 |
| Fuc/Xyla | 4 |
| Glc | 3 |
| a Not discriminated due to overlapping peaks. | |
Analysis of the eggshell amino acid composition
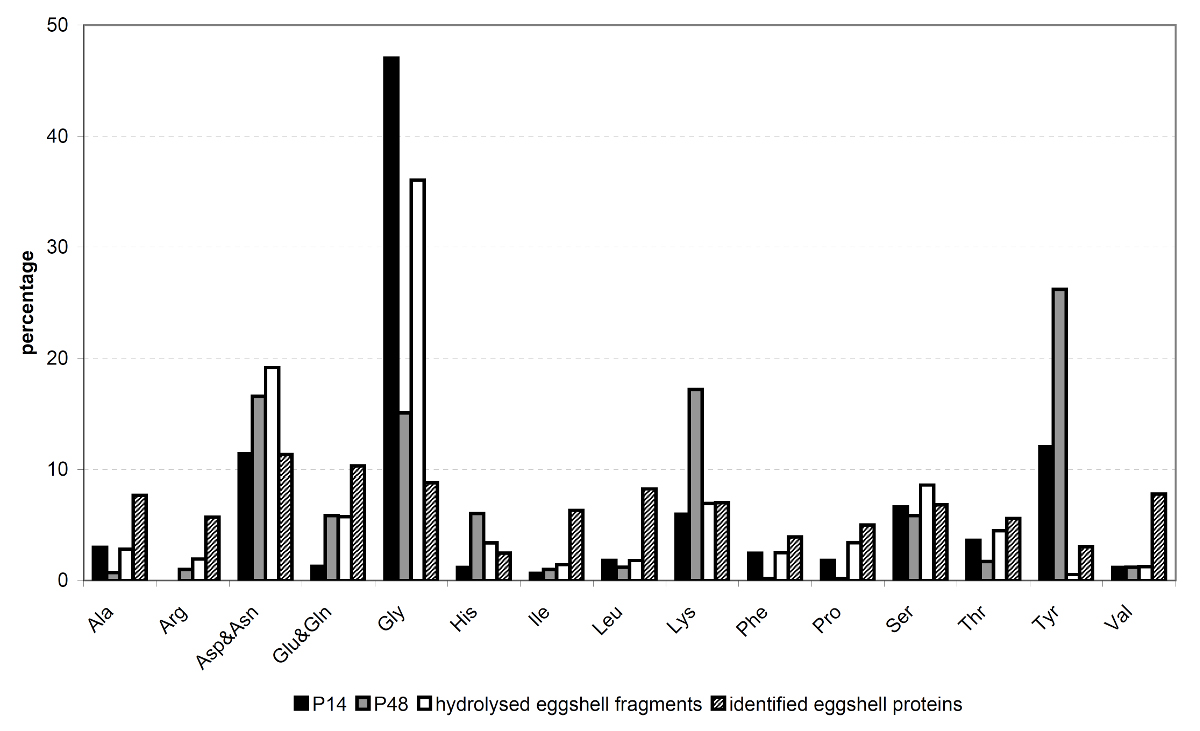
Putative eggshell proteins were identified in the late 1980s by searching for specific cDNAs that are only expressed in mature females and not in males, immature female worms, miracidia or cercariae. Examples of female-specific proteins that fulfill these criteria are p14 (14) and p48 (33). These putative eggshell proteins appeared to have a characteristic amino acid composition, containing either high glycine and tyrosine levels (p14) or high lysine and tyrosine levels (p48). Although these proteins are expected to be abundantly present in the protein skeleton of the eggshells, we did not identify these proteins in our proteomic analysis. Since p14 and p48 both contain high amounts of tyrosine residues, which are modified and cross-linked during the process of quinone tanning, identification of these proteins by peptide fingerprinting using MS is hampered by the post-translational modifications. Therefore, we performed amino acid analysis on the purified eggshell fragments (Fig. 2.6) to see whether it is likely that p14 and p48 were present in our sample, and hence are part of the insoluble protein skeleton of the eggshell. The amino acid composition of the purified eggshell skeleton was very similar to previous reports (24,150,195). High glycine levels (36%) suggest that p14 is indeed present in the purified eggshell fragments and is a highly abundant eggshell protein. In female worms, p14 mRNA, which is known as the F10 gene, is the most abundant transcript (128). Using immunostaining we were indeed able to confirm the presence of p14 in our samples of purified eggshells (Fig. 2.7).
Although p48 was expected to be part of the eggshell protein skeleton, our amino acid analysis did not reveal its presence. Lysine, an abundant amino acid of p48, was average in eggshell (6.8%) and tyrosine, another important amino acid of p48 could hardly be detected. Both of these amino acids are involved in quinone tanning and this process renders them unrecognizable as lysine and tyrosine.

Immune reactivity of the eggshell proteins
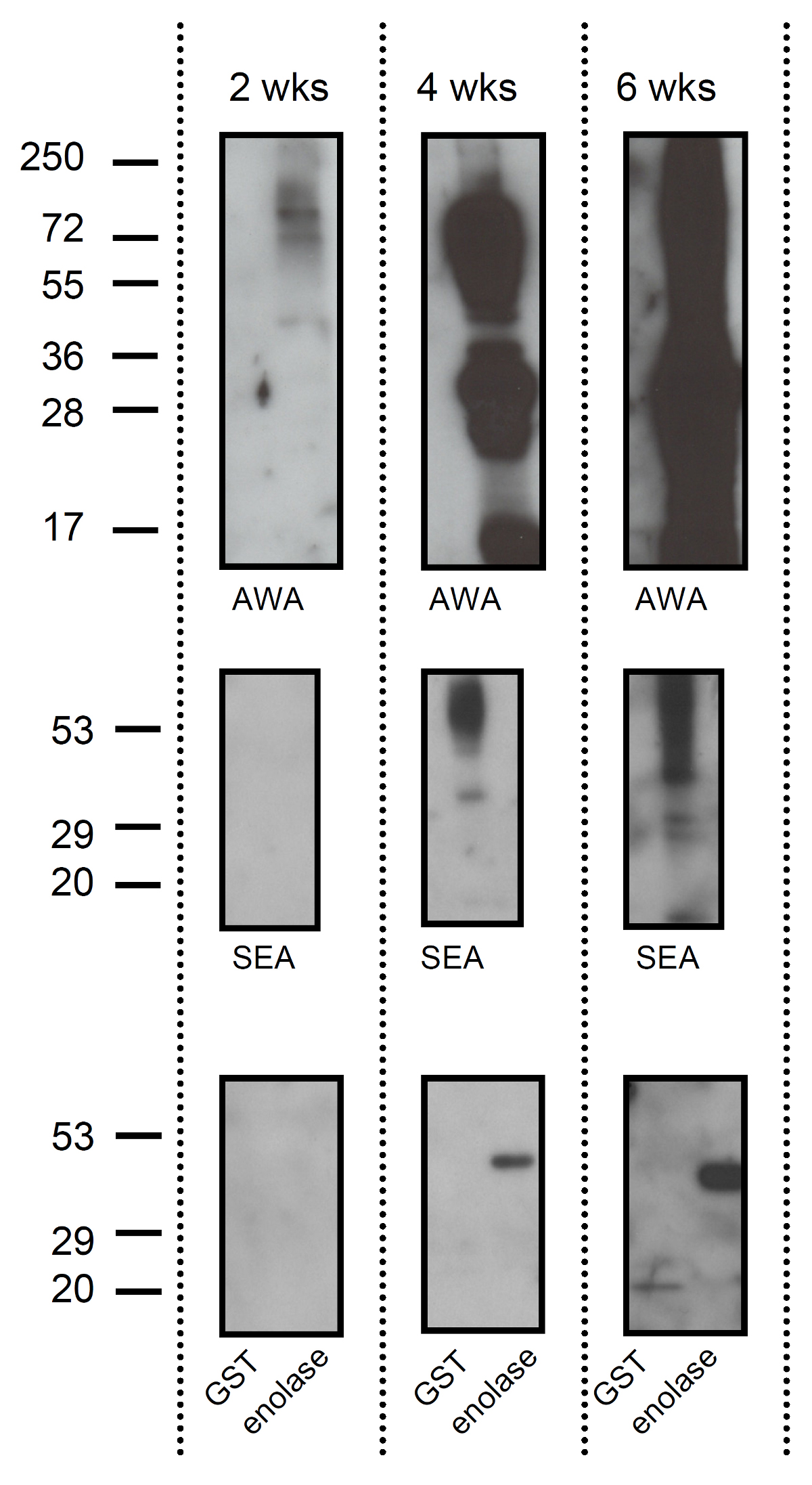
A number of the identified eggshell proteins such as GST, enolase and glyceralderhyde-3-phosphate dehydrogenase (GAPDH), are known to elicit an antibody response upon infection (122). Fig. 2.8 shows how GST and enolase, two of the identified eggshell proteins, stained positive on Western blots when incubated with sera from infected hamsters. Sera were taken from infected hamsters at 2, 4 and 6 weeks of infection. A positive stain for enolase was detected at 4 weeks of infection, which increased at 6 weeks of infection, when GST was also detected. In addition, antibody reactivity against SEA and AWA was tested. Like enolase, SEA started to stain positive at 4 weeks of infection and the signal increased at 6 weeks of infection. Antibody reactivity against AWA started earlier, at 2 weeks of infection and also increased during the course of the infection. Note that at 4 weeks of infection, worms do not produce eggs yet but enolase, a protein we demonstrated to also be present in eggshells, is already recognised by the serum. This demonstrates that enolase is exposed or secreted by developing worms before this protein is presented to the host via incorporation in the Schistosoma eggshell.
Discussion
The eggshell is the site of direct interaction between the highly immunogenic eggs of S. mansoni and the immune system of its host. Eggs are known to excrete immunogenic proteins (30,110), but it is likely that the proteins that form the eggshell also cause an immunological reaction. The exact protein composition of this rigid cross-linked structure was unknown. The aim of the current study was to identify the proteins that make up the cross-linked eggshell, the outer surface of the egg that interacts with the immune system.
In samples of the purified protein skeleton of eggshells we identified a collection of proteins, similar to those found in other proteome studies of S. mansoni (16,40,30,110,184). The variety of proteins we detected seems rather random (Table 2.1). Included are many enzymes involved in various metabolic processes, but for instance enzymes involved in crosslinking of the eggshell were not detected. Surprisingly, we also could not identify by MS either one of the putative eggshell proteins p14 and p48. We have two explanations for this. First, p14 and p48 are tyrosine-rich proteins, which means that it is likely that most of the peptides that are obtained after trypsin digestion will contain one or more tyrosines that have been modified to form cross-links in the quinone tanning process. Thus, tyrosines will not be detected and different side chains will add unknown and variable masses to the peptides which then cannot be identified by peptide mass fingerprinting. Second, in silico trypsin digestion of these putative eggshell proteins reveals that trypsin digestion leads to many very short peptides, which are very similar to each other (not shown). This is especially the case for p48, where trypsin digestion results in many pentapeptides of which peptide masses are under the detection limit of the mass spectrometer. Even when detected, such small peptides will not reach a 99.0% probability score for protein identification as they are too short to be specific and thus these peptides will not identify a protein using our criteria for protein identification. It will therefore be impossible to identify these putative eggshell proteins by LC-MS/MS, even if they are abundantly present in the eggshell.
Although p14 could not be identified in our MS analysis, using immunostaining we demonstrated the presence of this bona fide eggshell protein in purified eggshell fragments (Fig. 2.7). It is not just only present, but the extremely high glycine content (36%) of purified eggshell fragments detected by amino acid analysis indicates that p14 contributes largely to total eggshell. The glycine content of eggshell, of the identified proteins and of p14 enabled us to make a general estimation of this contribution of p14 to total eggshell. In the proteins that we identified by MS, average glycine levels were 8.2% of the 17 analysed amino acids, ranging from 4 to 25%. In p14, 47% of the amino acids were glycine. This leads to the following equation: [p14] × 47 + [rest] × 8.2 = 36, where [rest] = 1 - [p14], which demonstrates that p14 contributes to total eggshell for approximately 70%.
The set of proteins that contribute to the eggshell for the other 30% seems to be rather random. However, its average glycine level is significantly higher (7.7%) than the average of all 13,529 S. mansoni proteins available on Uniprot (4.8%) (P <0.001), while tyrosine (3.1% versus 3.4%) and lysine levels (6.8% versus 6.0%) are similar. Although glycine levels in identified eggshell proteins do by no means approach that of total eggshell or p14, its elevated levels in the identified eggshell proteins are remarkable. Apparently, there is a preference for proteins with higher glycine levels during cross-linking, although glycine does not contribute to quinone tanning.
While our analysis of the amino acid composition of eggshell indicated that p14 is present in the eggshell, such an analysis could not be used to estimate the presence of p48. Although p48 is rich in tyrosine, lysine and aspartic acid, none of these amino acids was abundant in our samples. As the amount of p48 in eggshell is known to be much less than p14 (33,93,102), tyrosine, lysine and aspartic acid levels of p48 are not abundant enough to feature in a mixed sample of total eggshell proteins. Furthermore, lysine and tyrosine that have been modulated to form cross-links by quinone tanning could not be traced back in the amino acid analysis after hydrolysation, resulting in very low detection of these amino acids.
The majority of the eggshell proteins identified by MS were non-structural proteins. Although these proteins were unexpected, immunoscanning microscopy of HSP70 validated our results. The identified proteins are not specific for the eggshells. Many identified proteins such as actin, tubulin, p40, HSP70 and glycolytic enzymes have also been identified by Cass et al. (2007)(30) as proteins secreted by S. mansoni eggs. In contrast, Mathieson and Wilson (110) found that these proteins are not present in secretions of eggs. Secreted or not, it is not likely that our samples were contaminated with proteins secreted by eggs because our eggshell fragments were cleaned extensively before proteomic analysis. The absence in our samples of proteins secreted by eggs is confirmed by the absence of IPSE/alpha-1 and omega-1, the two major proteins secreted by eggs. Similarly, contamination of our purified samples with other proteins originating from Von Lichtenbergs envelope, Lehman's lacuna or Reynolds' layer is just as unlikely due to the extensive pre-treatment.
The proteins identified in our purified fraction (Table 2.1; Fig. 2.7) are largely comparable with those of other egg fractions and even with those of other developmental stages of the parasite (16,40). The best explanation for the presence of these abundant cellular proteins, originating from surrounding vitelline cells, is that they happened to be around at the site and time of eggshell synthesis and were cross-linked to the major eggshell proteins. The absence of the two most abundant proteins secreted by eggs, IPSE/alpha-1 and omega-1, gives an indication of the timing of the cross-linking process. After production of the egg further maturation occurs, as the egg excreted by the female worm is still undeveloped and consists of an ovum and vitelline cells surrounded by the eggshell. Our results thus indicate that the actual formation of the eggshell including cross-linking of the proteins is most likely finished before the miracidium starts secreting proteins.
Although the observed incorporation of proteins originating from neighbouring vitelline cells may seem to be an unintended feature of eggshell production, the presence of these proteins may have immunological consequences. Many of the proteins we identified as part of the eggshell protein skeleton are known schistosome antigens such as p40, phosphoenolpyruvate carboxykinase (PEPCK) and thioredoxin peroxidase. These proteins induce cellular responses (3,200) or antibody responses (122). The immunogenicity of these usually intracellular proteins can now be explained as these proteins are part of the eggshell. As a consequence of these immunogenic properties, some of the eggshell proteins identified, such as GST and GAPDH, were proposed as vaccine candidates. However, the success of these vaccines in preventing infection was limited (133). This might now be explained by the fact that the vaccines target the eggs where the proteins are exposed and not the actual infection by the adult worms. In fact, vaccination with such antigens might even induce pathology as it enhances the immune response against the eggs, thereby enhancing granuloma formation and disease symptoms.
Altogether, our results demonstrate that the eggshell is not only composed of the specific eggshell proteins p14 and p48, but also includes proteins available at the site of eggshell production. Therefore we propose a new model of the Schistosoma eggshell. The main component of the eggshell is the putative eggshell protein p14. It makes up the majority of the eggshell. However, at the same time other proteins, whichever are available at the place and time of eggshell production, are also incorporated, albeit to a much lesser extent. Cross-linked together, all of these proteins make up a very rigid structure to protect the developing miracidium.
Acknowledgements
We thank Professor Phil LoVerde (University of Texas Health Science Center, San Antonio, Texas, USA) for providing us with the anti-p14 antibodies and for valuable comments on the manuscript. Niels Bohnen and Marion Schmitz are thanked for their contributions to experiments on the purification of eggshell.