Chapter 1
General introduction
Schistosomiasis
Blood-dwelling parasitic trematodes (flatworms) of the genus Schistosoma cause the disease schistosomiasis or Bilharzia. There are 5 different Schistosoma species that infect humans and many other infecting different mammals. Over 200 million people worldwide are infected with schistosomes, mainly of the species S. haematobium, S. mansoni and S. japonicum (37,197). The disease is endemic in tropical areas and endemicity is dependent on the presence of the intermediate host, an aquatic snail, in fresh surface water. There is a strong variation in epidemiology between regions and localities, depending on local determinants such as irrigation or draining canals and human sanitary conditions (74).
The life cycle of S. mansoni
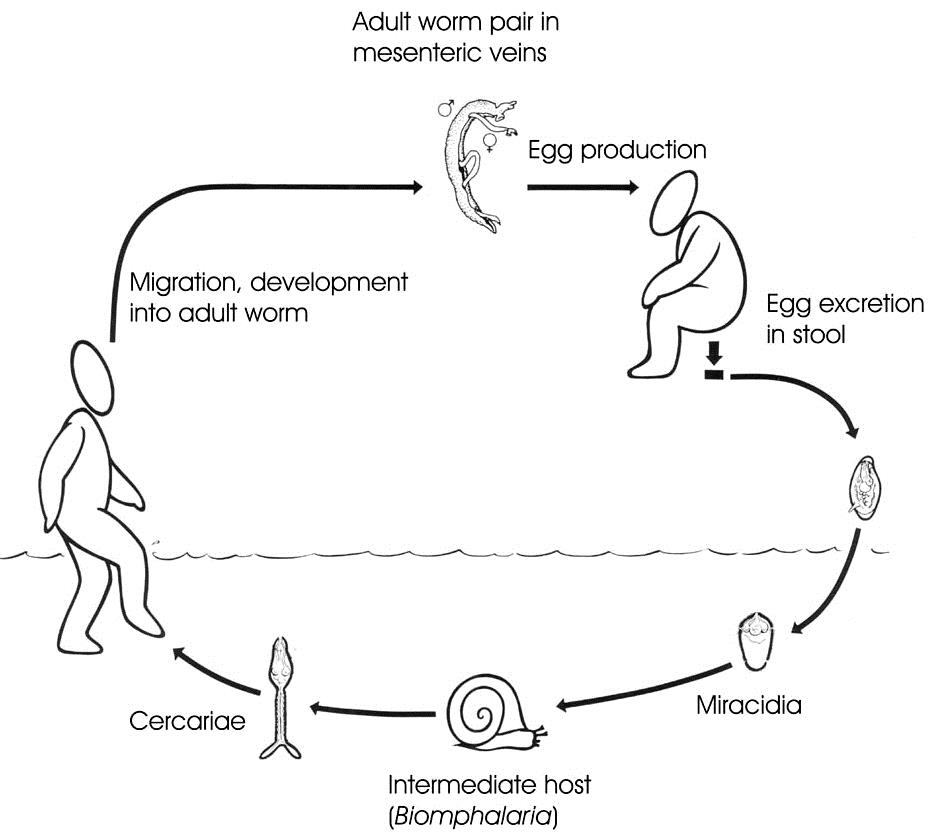
The life cycle of S. mansoni is depicted in Fig. 1.1. Infection of the human host occurs upon water contact, where larvae called cercariae are released from the snails and swim in the water in search for their mammalian host. They penetrate the skin. At this point, cercariae lose their tail and the cercarial body transforms into a migrating juvenile worm, a schistosomulum. In the circulation, schistosomula migrate to the lungs. In the case of S. mansoni schistosomula further migrate to the portal vein in the liver where male and female worms pair and mature before homing at their final destination, the mesenteric veins surrounding the gut. This occurs about six weeks after skin penetration (74,155).
In the mesenteric veins, adult worm pairs can reside for many years (37), feeding on blood and meanwhile producing 300 eggs per day (32). About half of the deposited eggs flow with the circulation and get trapped in the liver, where granulomas are formed around the eggs due to a strong immune response. The other eggs extravasate the vessel wall and pass through the intestinal wall in order to be secreted with the feces from about one week after being released by the female worm. It may take up to six weeks for the eggs to exit the host. However, eggs die within two weeks after oviposition (37), thus not all excreted eggs are viable.
Viable eggs that are released in water hatch and the larvae called miracidia can subsequently infect the intermediate host, the aquatic snail Biomphalaria glabrata or B. pfeifferi. Within the snail, massive clonal replication of the parasite, now sporocysts, occurs. About 5 weeks after infection, snails start to shed cercariae. Some of the cercariae will infect a human host.
Disease symptoms
Infections with S. mansoni often occur without notice. However, symptoms may be present in all stages of the infections (75).
The penetration of the skin can cause some local irritation called swimmer's itch. This usually does not last longer than a few hours. When seen, this is most often in primary infections, mainly in migrants and tourists. It is more often seen after infections with non-permissive schistosome species, such as Trichobilharzia (35,75).
Acute schistosomiasis, also called Katayama fever, can occur in primary infections with high numbers of migrating schistosomula (35). It results from a hypersensitivity T helper 1 T-cell (Th1) response against migrating juvenile worms. The symptoms are flu-like and may include fever, fatigue, myalgia and dry cough. At this stage, antibodies against schistosomes may be found, but stool and urine samples are negative for schistosome eggs. There is a marked eosinophilia and patchy infiltrates can be seen on chest radiography. Usually, symptoms disappear within a few weeks (35,75).
Abdominal symptoms caused by the migration and positioning of mature worms can occur at a later stage, when worms have matured and migrate to their final destination (74).
Schistosomiasis is most often apparent as a chronic disease. At this stage, much damage can be done of which some is irreversible. The major symptoms of disease are caused by the eggs that elicit a strong immune response. The severity of disease symptoms is dependent on the intensity and the duration of the infection as well as on host immune factors. In and around the intestine the immune reaction to eggs may lead to micro-ulcerations, pseudopolyps and microscopic bleeding causing abdominal pain, loss of appetite, diarrhea, anemia and malnutrition. The more serious disease symptoms are caused by eggs trapped in the liver. The immune reaction causes granulomas to form around the eggs. But it can also cause damage to the liver, leading to fibrosis of the liver, liver enlargement and portal hypertension. It is referred to as hepatosplenic schistosomiasis when portal hypertension leads to splenomegaly, portocaval shunting and/or external or gastrointestinal varices with the potential of fatal bleeding (74).
Ectopic schistosomiasis can develop in many tissues accidentally reached by worms and/or eggs. In advanced hepatosplenic schistosomiasis, portocaval shunts or portopulmonary anastomoses via the azygos vein may form as a consequence of portal hypertension. As a result, eggs may pass into the arterial circulation and get trapped in the lungs (75). Granulomas have also been found in the skin, adrenal glands, skeletal muscle, spinal cord and in the brain (27). The most severe form of ectopic schistosomiasis is neuroschistosomiasis, where eggs are present in the central nervous system (CNS). Cerebral involvement can be asymptomatic in immune patients with chronic hepatosplenic schistosomiasis, when eggs are scattered around the brain. Symptomatic cerebral involvement is more often seen in S. japonicum infection than in infections with other schistosome species. It usually starts within weeks after the infection (28) and may present with an acute or subacute onset of headaches, seizures, altered sensorium, motor weakness, focal neurological deficit, visual impairment or nystagmus, speech disturbances, and cerebellar symptoms (27).
Acute transverse myelopathy is most often caused by S. mansoni or S. haematobium. It is a severe disabling condition and is underdiagnosed in endemic areas (27). Transverse myelopathy due to schistosomiasis is more common in young adults, teenagers and children of the male sex. This may be explained by the higher exposure of male individuals to infected water during playing and working activities (63). The transverse myelopathy develops in an early stage of the infection, just after the start of egg deposition and often in the absence of other systemic symptoms (27). The eggs are deposited in situ after improper migration of adult worms to blood vessels of the central nervous system, usually equal or below T6, particularly at T11-L1 (63). Inflammatory and cellular immune reactions around the eggs mechanically suppress the spinal cord and cause neurological dysfunction. The main clinical symptoms are lower limb weakness that may be severe enough to prevent walking, lower limb pain, usually irradiating to the lower limbs with symmetrical or asymmetrical distribution, deep tendon reflexes abnormalities (hyper, hypo, and areflexia) and bladder, intestinal and sexual dysfunction. Usually, sensory symptoms and pain precede motor symptoms. The time between the initial symptoms and full neurological picture is often less than two weeks (28,63).
Diagnosis
Diagnosis of an S. mansoni infection is based on the microscopic detection of eggs in stool samples. The eggs of S. mansoni are relatively large, measuring around 140 - 60 μm, and have a characteristic lateral spine. The severity of an infection can be estimated by determination of the amount of eggs per gram feces using the Kato-Katz method (91), although there is a wide day to day variation in egg excretion (7,45). Microscopic detection of eggs in stool samples is a simple, cheap and quick method of detection However, sensitivity is low for infections with a small number of worms (44). Eggs can also be detected in material obtained from a rectal biopsy. Although a higher sensitivity can be achieved this way (75), rectal biopsies are not routinely done as this is invasive and costly.
There are dipstick tests that detect circulating cathodic antigen (CCA) or circulating anodic antigen (CAA) with monoclonal antibodies. The sensitivity of these tests is modest, but they have shown useful for screening, follow-up of chemotherapy and re-infection, control programmes and epidemiological studies (75).
More sensitive are the serological tests that detect the presence of anti-schistosome antibodies in blood using ELISA or indirect hemagglutination assays (IHA). Seroconversion usually occurs 4 to 8 weeks after infection (73,75). Although more sensitive, a major drawback is that test results may remain positive long after the infection is cleared (75). These tests are therefore very useful for detection of S. mansoni infections in travelers and other incidentally exposed individuals, but are much less useful in endemic areas.
Schistosome DNA can be detected in stool samples. With real-time PCR, the amount of DNA and thus the intensity of an infection can be quantified (180). However, due to costs and technical issues, PCRs are rarely performed routinely in low-income endemic areas.
Additional tests can be performed to determine pathology. Esophagal varices can be visualized by endoscopy or contrast radiography, while ultrasound, laparoscopy and wedge biopsy can reveal granulomas or periportal fibrosis (75). In ectopic schistosomiasis, imaging techniques such as CT or MRI scans may reveal lesions and edema for example around the spinal cord or in the brain. These findings are unspecific for schistosomiasis, but may be of additional diagnostic value (28,63).
Treatment and control
Praziquantel (PZQ) is the main drug used to treat schistosomiasis. It is cheap, safe and highly effective against all schistosome species. Unfortunately, eggs and juvenile worms are not affected by PZQ and the drug does not prevent reinfection (50,75). A treatment consists of one single oral dose in endemic areas where reinfection is likely. In travellers, two treatments with one month interval are given to eradicate all worms (207). In addition to the low costs and high effectiveness of PZQ, the side-effects of PZQ are mild and transient (50). They include nausea, vomiting, malaise and abdominal pain. But in heavy infections, massive worm shifts and antigen release after treatment may cause acute colic with bloody diarrhea (75). PZQ is effective within a few hours after intake (74). It causes tetanic contractions of adults worms as a result of a rapid influx of calcium ions, and causes disruption of the tegument, the worm's surface, after which worms are cleared by the immune cells (50).
Although PZQ treatment does not prevent reinfection, it may help to develop increased immunity to reinfection. Adult worms release antigens when they die inducing a humoral immune reaction that is partially protective against future infections (122). This indicates that immunity to schistosomes can be acquired (78,114), a finding which is further supported by the fact that the rate and intensity of infection in endemic areas generally decreases with age, i.e. children are more often and more heavily infected than adults. However, no successful vaccine has been developed so far (37).
For the treatment of neuroschistosomiasis, it is essential to administer corticosteroids in addition to PZQ (87). This is because the immune response following PZQ treatment can aggravate the neurological disorder. As corticosteroids are immunosuppressing, they reduce the inflammation around eggs and dying worms, thereby resolving or reducing neurological damage.
Host-parasite interactions
Immunology
The immune response in schistosomiasis follows a characteristic pattern. In the acute phase, schistosomula migrating through tissues induce a Th1 response. This response is characterized by elevated levels of the pro-inflammatory cytokines IFN-γ and interleukin (IL)-2 (76,135).
However, after about 8 weeks of infection, when worms have matured, have migrated to the mesenteric veins and have started egg production, a Th2 response becomes dominant. This response is characterized by high levels of the cytokines IL-4 and IL-5 and the presence of alternatively activated macrophages, a macrophage phenotype that usually occurs in the context of a Th2 cytokine environment (52). Furthermore, the Th2 response in schistosomiasis is accompanied by elevated levels of eosinophils and IgE. The elevation of IL-4 and IL-5 is linked to a down-regulation of the Th1 response, which is marked by a decrease in IFN-γ and IL-2 secretion (135). The switch from Th1 to predominantly Th2 is initiated by the deposition of eggs by mature female worms (76,135). IL-5 is needed for the development of eosinophilia but has no effect on the circulating levels of IgE (167). In contrast, IL-4 does increase IgE levels (136).
When the disease becomes chronic, which is around 12 to 16 weeks after infection, the immune response gets dominated by a regulatory T-cell response causing the immune suppression characteristic to chronic schistosomiasis (53,137). The granuloma sizes around the eggs now reduce. The induction of regulatory T-cells seems to be an important way of controlling over-vigorous immune responses during the course of chronic schistosome infection (53). IL-10, a regulatory cytokine, has an important role in tempering both Th1 and Th2 responses. Together with TGF-β, IL-10 redundantly reduces liver pathology by suppressing proinflammatory cytokine production (80,84).
Egg secretion products
As mentioned above, the majority of disease symptoms in schistosomiasis is due to cellular immune responses towards eggs trapped in the host tissues (75,76). Live mature eggs actively secrete proteins, some of which are heavily glycosylated. It has been shown that these glycans in SEA are responsible for the induction of a Th2 response from SEA (129). The major egg secretion proteins, the glycoproteins IPSE/alpha-1 and Omega-1 are both strong inducers of IL-4 production and Th2 development (59,164,178). IPSE/alpha-1 triggers basophils to produce IL-4 (164), whereas Omega-1 acts on dendritic cells to induce Th2 polarization (59,178). Omega-1 can induce Th2 responses even independently of IL-4 (59).
It is this characteristic Th2 response that induces granuloma formation around eggs. In addition, Th2 responses are involved in the development of naturally acquired resistance to reinfection with schistosomes (137). A T-cell reaction against eggs is also of major importance for the extravasation and excretion of eggs out of the host (49,54,90).
The eggshell
Not only the egg secretion products, but also the eggshell itself is immunogenic. The eggshell is the outer layer of the eggs and hence a direct site of interaction with the immune system. It is a hardened and tanned structure made from proteins cross-linked by quinone tanning, rendering eggshell intractable to protease activity. It is formed in the female reproductive tract, where the fertilized oocyte gets surrounded by vitelline cells that contain eggshell precursor proteins that are released upon contractions of the ootype (175). Tyrosinase activity causes cross-linking of the precursor eggshell proteins within single proteins as well as between neighbouring proteins. The resulting eggshell is protease resistant and very rigid, while the eggshell is at the same time very porous, enabling passage of egg secretion products.
The tegument
Adult worms are able to prevent an adequate immune response, allowing them to reside within the veins for many years. The outer surface of the schistosomes, which is called the tegument, is believed to play an essential role in this. This structure is unique to blood-dwelling trematodes and contains unique proteins and lipids (21,184). The tegument is composed of two closely apposed lipid bilayers with different properties on a layer of fused cells, the syncytium. The syncytium contains inclusion bodies from which the tegument is formed and maintained. The inclusions move apically and merge with the existing plasma membrane, where they release their content, which is then incorporated into the outer bilayer (82,83,113).
The bilayers mainly consist of phospholipids and large amounts of cholesterol. On a molar basis they contain more cholesterol than phospholipids (2). This large amount of cholesterol together with the high content of sphingomyelin and saturated (ether-linked) phospholipid species in the tegumental membranes results in a tight packing of the tegumental membranes, which renders them more rigid in physical terms.
The most abundant phospholipid classes in the tegumental membranes are phosphatidylcholine (PC) and phosphatidylethanolamine (PE) (151). This is also true for most eukaryotic membranes. However, the fatty acid composition of phospholipids in the tegumental membranes of schistosomes is different from that of the membranes of the blood cells of the final host (2). Among the fatty acids of the tegumental membranes are several unconventional fatty acid species that are absent in this host. As schistosomes do not synthesize fatty acids de novo and all their fatty acids originate from the host, schistosomes must be capable of modifying fatty acids. It has been shown that schistosomes can modify fatty acids by chain elongation and by the introduction of desaturations (22,118).
With its surface-enlarging folds, spines, and pits, the tegument appears as typical digestive-absorptive epithelium (70,81). Indeed, the tegument is essential for the absorption of nutrients (5,25,39,153). In addition, the tegument is needed for the uptake of cholesterol and other lipids (120,152). There is a low-density lipoprotein (LDL)-like receptor on the surface of the tegument (158), however, phospholipid or cholesterol uptake from LDL has never been demonstrated.
Since the tegument is the interface between the parasite and its host, it is likely to play a role in the protection of the worm against the defence mechanism of the host (111,112). It is possible that binding of LDL is a way to hide the worm from the immune system by coating it with host material and thus shielding its own antigens (205). Other possible immune evasion properties of the tegument are proteolytic degradation of host defence proteins, rigid membrane biophysical properties and a rapid turnover of the tegumental membrane (1). A possible mechanical function has been assigned to the typical spines that are present on the surface. They were proposed to prevent occlusion of the veins as blood can flow through them. In addition, the spines could prevent damage caused by complement.
Despite the fact that the tegument contains immunogenic proteins (17,184), it does not seem to elicit an immune response that lethally harms the worms. Other blood-dwelling flukes have similar tegumental structures, while it is absent in intestinal worms or free-living worms. This suggests that the tegument consisting of a double membrane is specially adapted for worm survival in the blood circulation of their host (113,199).
Hemostasis
Hemostasis is the process of stopping bleeds. It consists of both clot formation and wound healing. In the intact circulation, clotting is prevented by secretion of thrombomodulin, nitric oxide (NO) and prostaglandin by intact endothelial cells. In the case of vascular injury, blood loss is prevented by vasoconstriction and formation of a platelet plug followed by coagulation.
Platelet plug formation - primary hemostasis
Primary hemostasis involves the formation of a platelet plug at the site of injury in order to reduce blood loss through the injured blood vessel. It requires platelet activation, adhesion to the vessel wall and release of platelet granule content resulting in platelet aggregation. Platelets, by their receptor glycoprotein (GP) Ia/IIa adhere to collagen exposed through the damaged endothelium. This occurs within a few seconds of the injury. The process is stabilized by von Willebrand factor (VWF) which bridges GPIb and subendothelial collagen. Bound platelets get activated and degranulate. They release adenosine diphosphate and thromboxane A2, which promote platelet aggregation. In addition, platelets release serotonin and thromboplastin, which induces vasoconstriction and stimulates secondary coagulation respectively. Platelets aggregate using fibrinogen and VWF as connecting proteins.
The structure and the role of VWF in hemostasis
VWF is a 250 kDa protein that forms large multimers. It is made by endothelial cells and by megakaryocytes. In megakaryocytes, VWF is stored in α-granules that are later partitioned into platelets. In endothelial cells, VWF is stored in cytoplasmic granules called Weibel-Palade bodies. VWF is released upon platelet activation and endothelial damage and in response to stimuli including histamine, thrombin, fibrin, vasopressing, epinephrine, dopamine and nitric oxide (NO) (105,159).
Each VWF monomer contains a number of specific domains in the following order: D1-D2-D'-D3-A1-A2-A3-D4-B1-B2-B3-C1-C2-CK. The A1 domain binds GPIb, the only receptor for VWF on non-activated platelets. After binding to VWF, platelets get activated. On activated platelets, the GPIIb/IIIa (integrin IIb3) becomes surface exposed. This enables the platelets to also bind the C1 domain of VWF. The A1 domain further binds heparin and collagen. The A3 domain also binds collagen. Hence VWF can serve as a bridging agent between the platelet plug and the extracellular tissue matrix exposed by the damaged vessel wall (105,159).
The role of endothelium in hemostasis
Intact endothelium inhibits platelet aggregation and coagulation by release of thrombomodulin, NO and prostaglandin I2. Upon injury, subendothelial collagen gets exposed to the intravascular lumen and passing platelets bind to the collagen with their GPIa/IIa receptor and get activated. Damaged endothelium secretes VWF, which also binds and activates platelets. Hence, when primary hemostasis is initiated, it triggers secundary hemostasis, which comprises the proteolytic clotting factor cascade and results in fibrin fibers and thrombus formation. Tissue Factor (TF) is another initiater of secundary hemostasis. TF is stored in subendothelial cells annd released upon damage of the endothelium. Shear stress is also an other activator of endothelial cells and thereby an activator of clot formation (177).
Schistosomes and the hemostatic system
Although schistosomes live in the blood circulation, they rarely give hemostatic complications. This is remarkable for an object sizing 0.5 mm in diameter and 5 mm long (sizes of an adult worm pair) in the mesenteric veins, which have a diameter of 1 to 4 mm. Such an obstruction causes turbulence and shear stress, conditions that normally predispose the development of thrombotic events as a result of endothelial damage, activation of platelets and blood coagulation (103,177). Also the attachment of the adults to the vessel wall is expected to activate or damage the endothelium (58,130). Furthermore, schistosomes provide a foreign surface to blood, an event that is also commonly associated with platelet activation and thrombus formation. However, platelets do minimally adhere to adult schistosome or isolated tegument (203). In contrast, platelets adhere massively to eggshell. The adhered platelets showed extensive spreading (203). It is likely that the binding of platelets to eggshell is essential for egg extravasation from the circulation.
Thesis outline/scope
The protein composition of different developmental stages of S. mansoni has been subject of investigation of many other researchers (15,16,17,30,40,77,110,184). So far, detailed information on the protein composition of the eggshell was missing, while this structure is a long lasting site of direct interaction between the parasite and the host immune system. We analyzed the protein composition of the S. mansoni eggshell. Due to its close contact with the miracidium and host plasma and due to the rigid structure of the eggshell, several technical challenges needed to be dealt with in order to retrieve a clean and analytic fraction of the S. mansoni eggshell. The results are presented in chapter 2. In addition to the proteome, eggshell glycans were analyzed.
Chapter 3 describes the formation and composition of the eggshell with the new insights acquired from the study described in chapter 2. In this chapter, possible consequences of the eggshell structure and composition for its interaction with the host are discussed.
One of the interactions between the eggshell and the host is further investigated in chapter 4. As was previously described, platelets bind to eggshell and get activated (203). We here analyzed the direct binding of host plasma proteins and in particular of VWF to the eggshell of S. mansoni. We demonstrated that multiple plasma proteins bind eggshell and that VWF binds eggshell directly through the A1 domain. This led to our hypothesis binding of platelets, VWF and other plasma proteins is essential for the adhesion of eggs to the endothelium, which is the first step in extravasation from the circulation.
Chapter 5 reports the use of an immunologic assay to diagnose neuroschistosomiasis. As neuroschistosomiasis can rapidly progress into full paralysis with permanent nerve damage, it is of great importance to rapidly diagnose this condition. The therapy for neuroschistosomiasis with immunosuppressive corticosteroids can be detrimental in case of viral or bacterial causes of transverse myelitis. Therefore, it is important to exclude any other infectious cause of myelopathy and to be certain of the diagnosis neuroschistosomiasis. The described method is based on differential humoral immune responses to eggs and worms in plasma and in cerebralspinal fuid (CSF). Several other possible molecular and immunologic methods were also evaluated for their usefulness in the diagnosis of neuroschistosomiasis.
The phospholipid composition of the tegumental membranes were analyzed and compared to whole worm and to mammalian cellular membranes. The results are described in chapter 6. In addition, culture supernatants and blood plasma of infected hamsters were analyzed for schistosome lipid secretion products. These analyses showed that tegument contain tegument-specific phospholipids of which species composition is considerably different from whole worm or host cells. These tegument-specific phospholipids could not be detected in serum of infected hamsters nor in culture supernatant. However, this does not necessarily imply that they are not secreted by schistosomes in situ in the host.
Chapter 7 summarises the conclusions of the work described in this thesis and possible implications of the results are discussed.